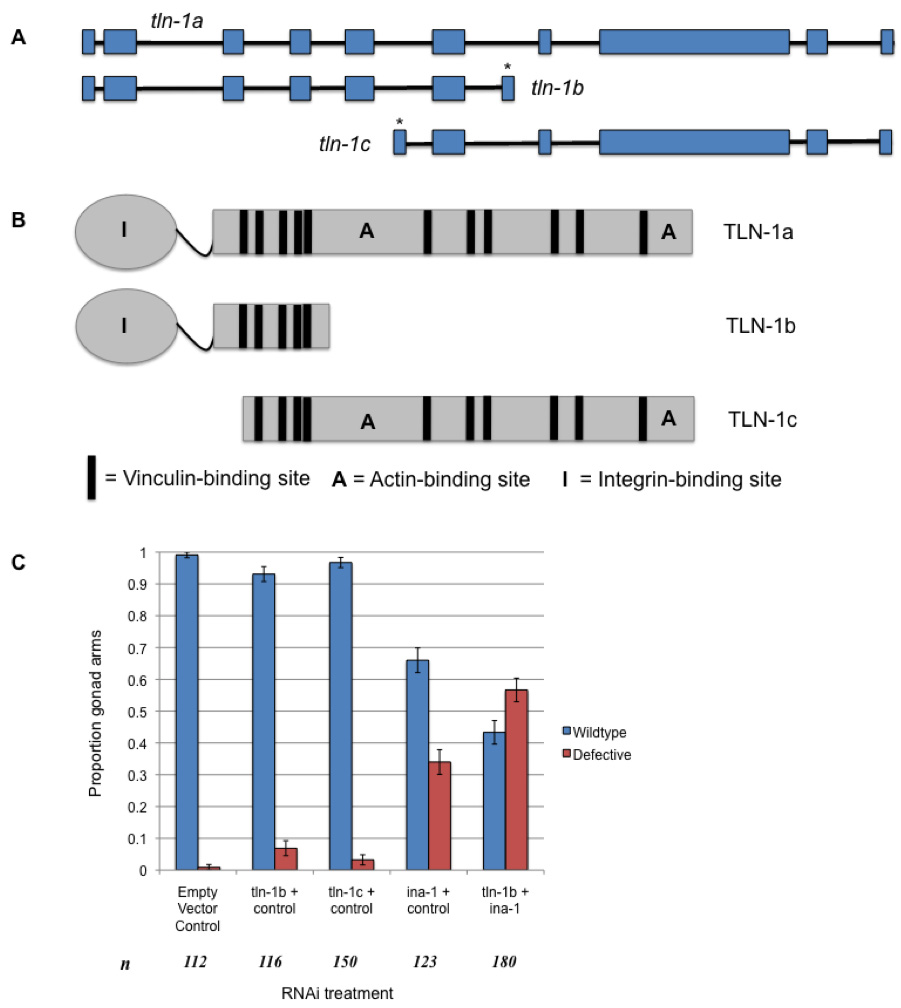
Talin localizes to cell adhesion sites, where it binds integrins, actin and vinculin (Petit and Thiery, 2000). In C. elegans, tln-1/talin is necessary for muscle cell attachment to the basal lamina via dense bodies (Lecroisey et al., 2007) and for gonad morphogenesis, which is driven by distal tip cell (DTC) migration (Wong and Schwarzbauer, 2012). In addition to the full-length transcript (tln-1a), two shorter splice isoforms (tln-1b, tln-1c) of the tln-1 transcript are predicted in C. elegans (Fig. 1A). Insights gained about specific tln-1 splice isoforms have implications for the functional profile of talin and roles of its various domains, which enhances our understanding of the mechanisms behind cell attachment and migration.
We confirmed tln-1b and tln-1c transcripts in N2 C. elegans cDNA through RT-PCR using primers targeted to their predicted unique exon sequences, indicating that they are transcribed (Fig. 1A). Based on conserved domain searches and sequence alignments, we identified domains encoded by the predicted amino acid sequences of tln-1b and tln-1c. TLN-1b contains the integrin-binding head domain and at least one vinculin binding site in the rod, while TLN-1c contains multiple actin and vinculin binding sites in the rod (Fig. 1B).
Knockdown of tln-1a using an RNAi construct that also targets tln-1c causes paralysis and DTC migration defects (Cram et al., 2003). To determine the individual roles of tln-1b and tln-1c, we performed RNAi feeding experiments using novel RNAi constructs that specifically targeted either tln-1b or tln-1c (Fig. 1A). tln-1b RNAi caused progressive total body paralysis in rrf-3(pk1426) nematodes: 97% of late-L4/adult animals were paralyzed by 55 hours of RNAi treatment (n=178). We next examined the localization of PAT-2/a-integrin using the JE2222 strain that carries the PAT-2::GFP transgene (Meighan and Schwarzbauer, 2007). Following tln-1b RNAi and paralysis, 66% of nematodes displayed aberrant muscle cell shape and PAT-2 localization (n=44), while 86% of nematodes showed disrupted actin filamentation (n=49). This suggests that tln-1b is important for muscle cell shape, PAT-2 localization, and cytoskeletal attachments. RNAi against tln-1c did not cause paralysis or muscle morphology defects.
Although loss of either tln-1b or tln-1c did not affect gonad morphology, double RNAi knockdown of tln-1b and ina-1/a-integrin produced a significantly greater proportion of defective gonad arms (p<0.05), including defects in migratory direction and early cessation, when compared to ina-1 reduction (Fig. 1C). The TLN-1b protein may interact with the INA-1 receptor during DTC migration. No synergistic effects were observed upon tln-1c + ina-1 double RNAi knockdown.
Our analyses indicate that tln-1b is important for muscle function and DTC migration, while specific functions of tln-1c remain to be elucidated. These findings suggest that multiple isoforms perform the biological roles of C. elegans talin and that the talin head domain is especially important for these processes. We propose that the structural simplicity of TLN-1b and its potential for strong integrin binding may be necessary for integrin clustering during adhesion formation, displacement of TLN-1a during adhesion disassembly, or integrin activation during dense body protein accretion, thus making it relevant for cell migration and muscle attachment.
Figures
References
Cram EJ, Clark SG, and Schwarzbauer JE. (2003). Talin loss-of-function uncovers roles in cell contractility and migration in C. elegans. J. Cell Sci. 116, 3871–3878.

Lecroisey C, Ségalat L, and Gieseler K. (2007). The C. elegans dense body: anchoring and signaling structure of the muscle. J. Muscle Res. Cell Motil. 28, 79–87. 
Meighan CM, and Schwarzbauer JE. (2007). Control of C. elegans hermaphrodite gonad size and shape by vab-3/Pax6-mediated regulation of integrin receptors. Genes Dev. 21, 1615–1620. 
Petit V, and Thiery JP. (2000). Focal adhesions: structure and dynamics. Biol. Cell 92, 477–494. 
Wong M, and Schwarzbauer JE. (2012). Gonad morphogenesis and distal tip cell migration in the Caenorhabditis elegans hermaphrodite. Wiley Interdiscip. Rev. Dev. Biol. 1, 519–531. 
Articles submitted to the Worm Breeder's Gazette should not be cited in bibliographies. Material contained here should be treated as personal communication and cited as such only with the consent of the author.
Leave a Reply
You must be logged in to post a comment.