2 Neuroscience Department, Oberlin College, Ohio
3 Neuroscience Program, University of Illinois Urbana-Champaign
Figures


Browser extensions are small software programs that enhance the user’s online experience by creating greater browser functionality. Here, I present a Caenorhabditis elegans specific extension for Google Chrome browsers. The software is built from four files: 1) an HTML file called celegansChrome.html that generates the user interface; 2) a JavaScript file called celegansChrome.js that contains objects of C. elegans gene data; 3) a JSON file that contains a manifest of the program contents called manifest.json; and 4) a small image file of a C. elegans hermaphrodite called icon.png that gets loaded into the browser toolbar. The software is freely available at the Google Chrome Web Store by simply clicking the add to Chrome link to load the extension into the browser:
chrome.google.com/webstore/detail/celeganschrome/ipnlfcanpnhkhljmminidjndijjmheib
The user can enter a gene common name (e.g., egl-4) and the Chrome extension can provide the user with C. elegans gene data from any website or even while off-line. Often, it’s convenient to retrieve linkage data or interactome data for a specific C. elegans gene while browsing different webpages or reading journal articles online – this extension provides a frictionless interface for the user to retrieve these data without having to open a new tab and search another website or paper for these data. Currently, the extension provides the user with linkage data for C. elegans genes, gene interaction data, human othology data to a C. elegans gene, and also a gene overview using the WormBase (Harris et al., 2014) RESTful API (only this feature requires internet connectivity). The linkage data loaded into the C. elegans Chrome extension was mined from WormMart WS220 (www.wormbase.org/), while the human ortholog data is from the InParanoid program database (Sonnhammer and Ostlund. 2015). The interaction data were downloaded from the WormBase version WS237 ftp site, and the overview of gene data relies on the WormBase RESTful API. The WormBase RESTful API provides access to gene data by creating a URI using standard HTTP requests that is unique to each gene – the unique identifier being a WBID. The C. elegans Chrome extension takes as input the C. elegans gene common name and maps it to the corresponding WBID to generate the unique URI.
This simple browser extension operates as a client-side program (with the exception of the overview option), and thus provides very fast and seamless data retrieval for C. elegans gene data while browsing any site online.
I started my independent research group from August 2014 in the Department of Biology at the University of Miami in Coral Gables, Florida. Our fundamental goal is to understand how neurons communicate in circuits to establish an appropriate level of activity that produces a robust, stable behavior. Our approach is to analyze in detail a model neural circuit that controls egg-laying behavior in C. elegans. We are taking advantage of the optical clarity and powerful genetics in this experimental system to literally watch the activity of every cell in the circuit in behaving animals using fluorescent Ca2+ reporters, and also to manipulate their activity using optogenetic tools. Using mutations and transgenes to discover and alter molecular signaling events between cells, we are determining how the complex pattern of activity in a circuit creates a coherent, regulated behavior. We expect these studies will reveal general principles of neurotransmitter signaling and neural circuit function with applications to understanding the human nervous system and its dysfunction in disease.
Website: http://www.as.miami.edu/biology/people/faculty/kevin-collins/
Ph.D. student and Postdoc positions are currently available. To apply, please send a CV and description of research interests to kcollins@bio.miami.edu
Many species of nematodes are known that are pathogenic as well as toxigenic for plants and many of them live as a parasite on the host plant causing severe crop damage and economic loss (Burglin et al., 1998). Although chemicals are available to control the harmful nematodes, they have their own set of deleterious effects such as decrease in soil fertility, impact on non target beneficial fauna, decrease in crop yield, and toxic effects on farmers. As fungi are known to have nematocidal activity (Khan et al., 2003), they may be used as a potential biological control agent.
In order to examine the effect of extra-cellular fungal extract as a nematicidal agent, C. elegans was used as a model system. In this study, we choose two Indian fungal isolates: (1) Aspergillus terreus, and (2) Fungal sp. US14, for its nematicidal potential on C. elegans. Fungal sp. US14 showed only 96% sequence similarity with Purpureocillium lilacinum with regards to its internal transcribed spacer 1 (ITS1), 5.8S ribosomal RNA gene and internal transcribed spacer 2 (ITS2) sequences. So, it might be a new species and was deposited at NCBI in the name of Fungal sp. US14 (NCBI accession number-JN802258.1).
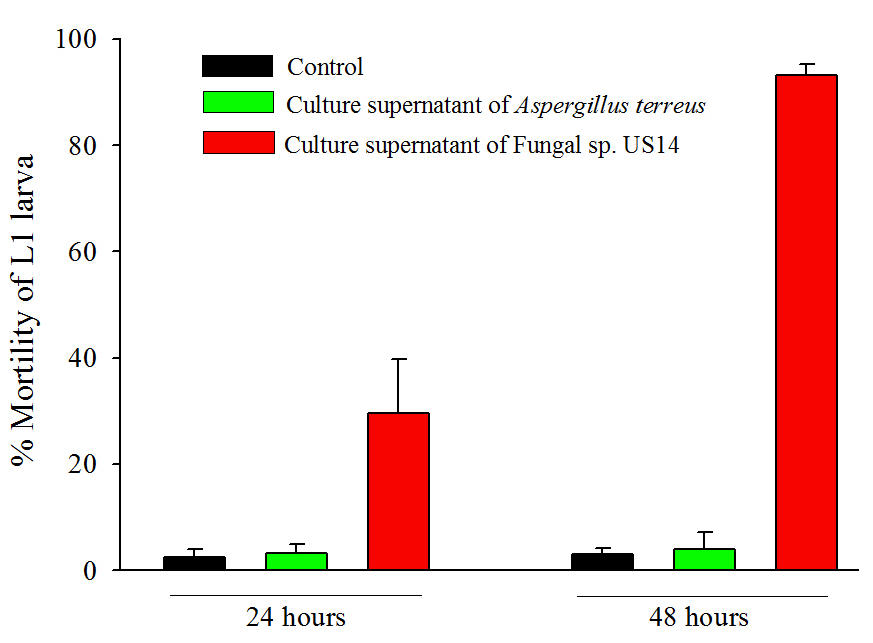
We carried out mortality assays on C. elegans using seven-day old grown-culture supernatant (extra-cellular culture filtrate) of both fungi. Synchronised L1 larvae of wild-type C. elegans were transferred with and without fungal supernatants, followed by incubation at 15oC. Mortality % of larvae was determined up to 48 hours by counting the dead larvae in the presence of fungal supernatant as compared to control animals which were grown in M9 buffer. We counted the number of dead larvae at 24 hours and 48 hours. The fungal supernatant of US14 killed 29±10.5% larvae at 24 hours and 93±1.9% at 48 hours (p<0.0001) compared to control larvae. The fungal supernatant of Aspergillus terreus did not kill larvae (Figure 1), and was almost similar to the control assay, suggesting its nonpathogenecity towards C. elegans.
Some of the study shows that the nematicidal activity of the fungus may be attributed to the presence of chitinase, protease, and lipases in the culture supernatant. The chitinase activity destroys the cuticle of nematodes and causes mortality (Miller and Sands, 1977). Furthermore, chitinase activity of nematophagous fungi has been previously correlated with their pathogenicity to some plant-parasitic nematodes (Stirling and Mankau, 1979). All together, the Fungal sp. US14 appeared as a promising candidate for biological control of nematodes.
Bürglin TR, Lobos E, and Blaxter ML. (1998). Caenorhabditis elegans as a model for parasitic nematodes. Int. J. Parasitol. 28, 395-411. 
Khan A, Williams K, and Nevalainen H. (2003). Testing the nematophagous biological control strain Paecilomyces lilacinus 251 for paecilotoxin production. FEMS Microbiol. Lett. 227, 107-111. 
Miller, PM and Sands, DC. (1977). Effects of hydrolytic enzymes on plant-parasitic nematodes. J. Nematol. 9, 192-197. 
Stirling, GR and Mankau, R. (1979). Mode of parasitism of Meloidogyne and other nematode eggs by Dactylella oviparasitica. J. Nematol. 11, 282-288. 
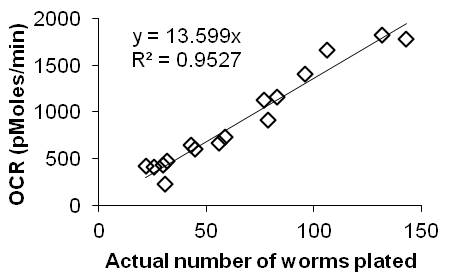
This is a protocol for the determination of oxygen consumption rate (OCR) in living C. elegans using the Seahorse apparatus with 24-well plates. OCR provides a measurement of respiration, which can be altered by genetic or pharmacologic manipulation of the mitochondrial respiratory chain. Although the Seahorse was originally designed for use with ex vivo cells, it can readily be used in vivo with living C. elegans by following our simple procedure,2 described here for the analysis of four samples side-by-side.
Synchronize each of the four samples of worms. We allow 200 adults to lay eggs for 4 h on a 10 cm plate by picking 200 adults to a 10 cm plate, kill the adults, then study the F1s as day 1 adults. Program the Seahorse Bioscience XF24 Extracellular Flux Analyzer with the following protocol: 1- calibrate probes; 2- loop 10 times; 3- mix 2 min; 4- time delay 2 min; 5- measure 2 min; 6- loop end. Furthermore, the internal heater should be switched off to allow the instrument to be close to room temperature. Hydrate the probes in the extracellular flux assay kit by adding 1 ml of Seahorse calibrant solution to each well and incubating overnight at 37 oC . Load this into the Seahorse on the day of the experiment for calibration.
Rinse the worms off the plate and wash three times using gravity separation in M9 and resuspend each washed sample in 2 ml. Get a rough estimate of the concentration of worms by pipetting 5 drops of 20 µl each per sample onto an empty dish and counting under the dissecting scope. There should be about 2 worms per drop. In the Seahorse assay plate, place 500 µl M9 into four blank wells. At least 30 min after the worms were first rinsed, pipet the four worm samples into a total of 500 ul M9 in each of the other wells with between 25 and 150 worms per well and five replicates of each sample. We use a low retention tip cut to create a wider opening, and found that picking worms into the wells with a platinum wire leads to unstable results.
When prompted by the instrument, replace the calibration plate with the worm plate. At the end of the run, count the actual number of worms per well; for this purpose, we capture images on a camera-fitted dissecting scope. Average the background-corrected OCR (pMol O2/min) across the 10 time-points for each well, then normalize the OCR to the actual number of worms in each well. Calculate the average and standard deviation of OCR per worm across the five replicates for each sample. A moderate amount of variation is expected between time-points, wells, plates, and days: we therefore use a minimum of 10 time points of 5 wells, and on at least 2 plates and/or days, to get a meaningful result.
1 For a more detailed protocol with additional tips, please contact Beverley at Beverley.M.Dancy@gmail.com.
2 We acknowledge other labs using Seahorse for C. elegans who provided some tips when we began this work, including Laurent Mouchiroud in Johan Auwerx’s lab at Ecole Polytechnique Fédérale de Lausanne, David Raizen at University of Pennsylvania, and Paul Brookes at University of Rochester Medical Center.
3 We thank José Antonio Mari (ja_mari@yahoo.com) for putting together the graphics in Figure 1.