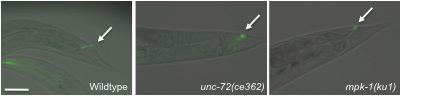
In its natural environment C. elegans encounters many bacterial pathogens and is capable of mounting a conserved immune response to protect itself from these infections. Microbacterium nematophilum (M. nematophilum) is a coryneform gram positive bacteria which binds to the post anal cuticle and colonises the rectal opening of C. elegans. This leads to a chronic, non-lethal infection characterised by gut distension, constipation and an innate immune response that includes swelling of the post-anal region known as the deformed anal region (DAR) phenotype (Hodgkin et al., 2000). M. nematophilum binding to the rectal opening can be visualised using the nucleic acid stain SYTO13 (Invitrogen), which shows green fluorescence when excited at 488-491 nm (Figure 1). Several groups including our own have used this nucleic acid stain to detect the presence of M. nematophilum colonisation in the rectal opening of animals that display the bacterially unswollen (BUS) phenotype following infection (Figure 1) (Nicholas and Hodgkin, 2004; McMullan et al., 2012).
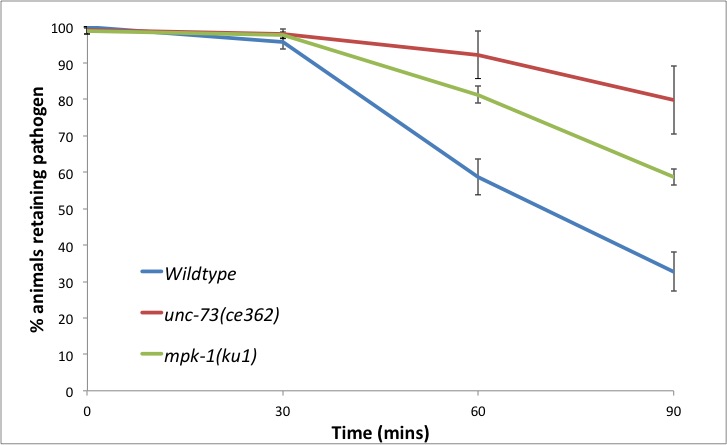
An important mechanism of the host defence system to combat and resolve infections is the ability to clear pathogens. We have utilised the easy detection of SYTO13 to follow the ability of C. elegans to clear M. nematophilum from its rectal opening. By scoring the number of animals in a population that show green fluorescence in the rectal opening at 30 minute time points we can see a decrease in M. nematophilum adherence over time (Figure 2). This reflects loss of M. nematophilum attachment rather than fluorescent decay or loss only of the SYTO13 stain, as re-incubation of the animals with fresh SYTO stain at a 90 minute time point results in the same number of animals with green fluorescence.
Bus mutants which still show pathogen binding, such as mutants in the EGL-30–UNC-73–RHO-1 pathway (McMullan et al., 2012) show a corresponding decrease in their ability to clear the SYTO13 positive M. nematophilum from the rectal opening (Figure 2).
This is the first demonstration of the positive role of the DAR phenotype in aiding pathogen clearance during infection and confirms that the DAR phenotype acts as part of the protective response. This method of quantifying M. nematophilum clearance could be applied to other pathogens where GFP labelled variants cannot be generated making it a simple method to assay the effect of mutations on an animal’s ability to clear infection.
Method: Adult animals were transferred from OP50 plates to infection plates seeded with 10% M. nematophilum and the progeny grown to L4-adult stage.
SYTO13 labeling was performed as described previously (Nicholas and Hodgkin, 2004) except that after a 60 minute incubation with SYTO13 10-20 µl of settled, stained worms were transferred to NGM plates lacking food. A ring of 150 mM Copper Sulphate, 2% SDS was used to prevent animals escaping from the plates.
The number of animals colonized by SYTO13 positive M. nematophilum was scored using a Nikon SMZ1500 microscope with GFP filter.
Acknowledgements:We thank Stephen Nurrish (University College London) for suggestions in setting up this assay. We thank Jonathan Hodgkin for M. nematophilum strains.
Figures
References
Hodgkin J, Kuwabara P, and Corneliussen B. (2000). A novel bacterial pathogen, Microbacterium nematophilum, induces morphological change in the nematode C. elegans. Curr. Biol. 10, 1615–1618. 
McMullan R, Anderson A, and Nurrish S. (2012). Behavioral and immune responses to infection require Gαq-RhoA signaling in C. elegans. PLoS Pathog 8, e1002530. 
Nicholas H, and Hodgkin J. (2004). The ERK MAP kinase cascade mediates tail swelling and a protective response to rectal infection in C. elegans. Curr. Biol. 14, 1256–1317. 
Articles submitted to the Worm Breeder's Gazette should not be cited in bibliographies. Material contained here should be treated as personal communication and cited as such only with the consent of the author.
Leave a Reply
You must be logged in to post a comment.