2Laboratory of Developmental Biology, Rutgers Center for Vector Biology, Rutgers University, New Brunswick NJ
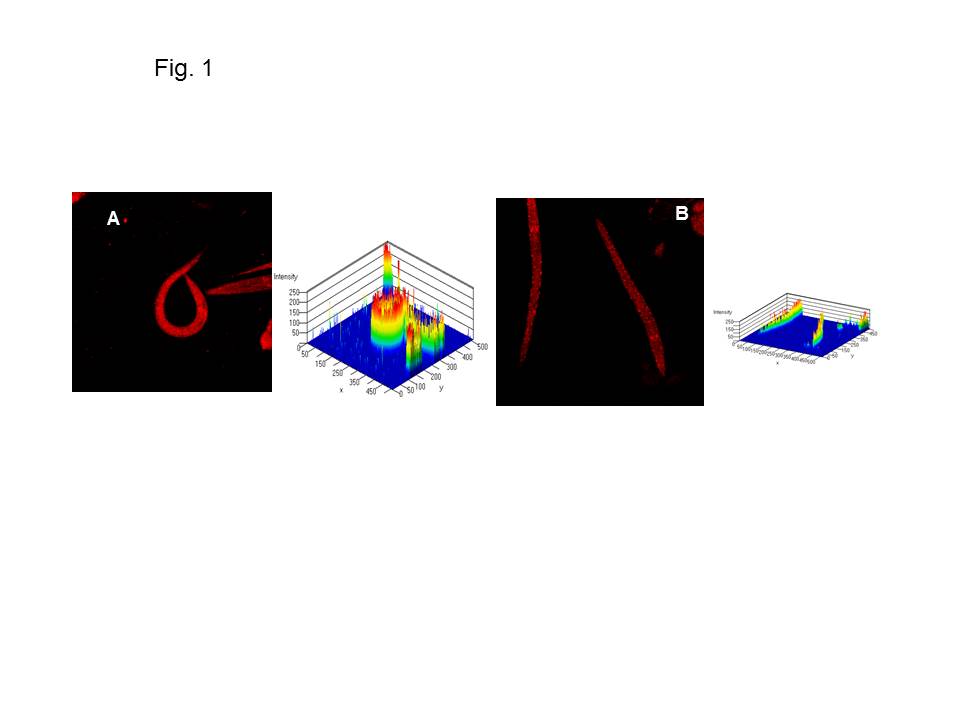
Fatty acid biosynthesis and oxidative degradation play a key role in lipid metabolism and their homeostatic balance controls energy storage. Its deregulation and dysfunction is linked to a wide range of disease risk factors or clinical manifestations from excessive deposit (obesity) to extreme lipodystrophy (cachexia). Cancer-cachexia is a complex metabolic condition in which disease-associated metabolic changes lead to a severe loss of skeletal muscle. Alterations in mitochondrial content, morphology, and function lead to several muscle-wasting conditions. Mitochondrial dysfunction has been reported in mouse models of cancer cachexia (Constantinou et al., 2011; Fermoselle et al., 2013). We have shown that one of the three Krüppel-like factors in C. elegans, KLF-3, a highly conserved homologue, possessed an important function in lipid metabolism probably via its transcriptional regulation (Zhang et al., 2009; Zhang et al., 2011; Zhang et al., 2013). klf-3 regulates beta-oxidation of long-chain fatty acids (Zhang et al., 2011) and impacts on the biosynthesis and mobilization of triglycerides (Zhang et al., 2013). We investigated if klf-3 influenced mitochondrial activity to modulate FA beta oxidation. We analyzed mitochondrial proliferation in synchronized populations of both wild type and klf-3 mutants using a modified Mito Tracker staining protocol (Sherwood et al., 2005). We found that mitochondrial content in the wild-type worm was notably high (Figure 1A), whereas klf-3 elimination reduced the mitochondrial content in the mutant worm (Figure 1B). Our data suggests that KLF-3-associated lipid accumulation in the mutant affects mitochondrial contents, leading to its dysfunction and reduced catabolism.
Recently we explored whether there is connection between fat metabolism dysfunction, reduced mitochondrial content, and muscle-wasting conditions. We characterized a joined deletion (rh160) of klf-3 and its downstream F54H5.3 gene in C. elegans strain NJ701. Through DNA sequencing, we determined the exact breakpoints of this deletion, which spans several kb of genomic sequence. RNA interference of F54H5.3 in wild-type N2 worms revealed a partial embryonic lethality and defective reproduction. However, F54H5.3 RNAi on the background of klf-3(ok1975), a smaller deletion that affects only klf-3, alleviated the fatty phenotype of the ok1975 mutant but recapitulated the severe muscle-wasting phenotypes associated with klf-3(rh160). Together, our data suggest that klf-3 and F54H5.3 interact in the same epistatic genetic circuit and potentially form an operon-like structure to coordinate their joint regulation in lipid metabolism. These observations bear novel implications for both obesity and type 2 diabetes and cachexia in cancer patients. Although our data is preliminary it does suggest a link between mitochondrial dysfunction, fat metabolism and cancer-cachexia. Our study may provide a simpler animal model to deepen our understanding of their underlying mechanisms.
Figures
References
Constantinou C, Fontes de Oliveira CC, Mintzopoulos D, Busquets S, He J, Kesarwani M, Mindrinos M, Rahme LG, Argilés JM, and Tzika AA. (2011). Nuclear magnetic resonance in conjunction with functional genomics suggests mitochondrial dysfunction in a murine model of cancer cachexia. Int. J. Mol. Med. 27, 15-24. 
Fermoselle C, García-Arumí E, Puig-Vilanova E, Andreu AL, Urtreger AJ, de Kier Joffé ED, Tejedor A, Puente-Maestu L, and Barreiro E. (2013). Mitochondrial dysfunction and therapeutic approaches in respiratory and limb muscles of cancer cachectic mice. Exp. Physiol. 98, 1349-1365. 
Sherwood DR, Butler JA, Kramer JM, and Sternberg PW. (2005). FOS-1 promotes basement–membrane removal during anchor-cell invasion in C. elegans. Cell 121, 951-962. 
Zhang J, Yang C, Brey C, Rodriguez M, Oksov Y, Gaugler R, Dickstein E, Huang CH, and Hashmi S. (2009). Mutation in Caenorhabditis elegans Krüppel-like factor, KLF-3 results in fat accumulation and alters fatty acid composition. Exp. Cell Res. 315, 
Zhang J, Bakheet R, Parhar RS, Huang CH, Hussain MM, Pan X, Siddiqui SS, and Hashmi S. (2011). Regulation of fat storage and reproduction by Krüppel-like transcription factor KLF-3 and fat associated genes in Caenorhabditis elegans. J. Mol. Biol. 411, 537-553. 
Zhang J, Yang C, Brey C, Rodriguez M, Oksov Y, Gaugler R, Dickstein E, Huang CH, and Hashmi S. (2009). Mutation in Caenorhabditis elegans Krüppel-like factor, KLF-3 results in fat accumulation and alters fatty acid composition. Exp. Cell Res. 315, 2568-2580. 