View the full size version.
Front Cover: Wild Types

View the full size version.
Protein extraction from C. elegans using sea sand
Making mini scale extracts from C. elegans for visualizing proteins and RNA has been difficult and inefficient due to the nematode’s sturdy outer shell, the cuticle. Boiling and sonication have been the methods of choice with some success, but both have their limitations. Boiling nematodes irreversibly denatures proteins, while sonication produces low, inconsistent yields. Even an extended period of sonication leaves many intact and halved nematodes in the extract, which renders biochemical experiments rather challenging and erratic. Boiling nematodes in SDS sample buffer provides an efficient way to visualize proteins on SDS-page protein gels, but certain proteins do not enter the gel due to the heat treatment. Here, we show a quick and efficient method of worm grinding with clean sea sand. The method is fast and high-quality extracts can be obtained in an Eppendorf tube for protein gel and RNA extractions. If desired, the sand can be separated from the extract and the clean extract can be obtained in a minimum amount of time.
Method: The method is optimized for 40 to 100 μL of clean worms. The worms should be totally cleaned from the bacteria with two to three washes with double distilled water (0.01% Triton X-100) in a clean Eppendorf tube prior to lysis. Extra water should be gently removed to make sure that addition of the 2X sample buffer yields the appropriate dilution of buffer for sand grinding. A small scoop of 30 to 40 μL volume of washed clean sand (Fisher Chemicals catalog #14808-60-7) should be added to the worm pellet. 2X LDS* (Lithium Dodecyl Sulphate) sample buffer with protease inhibitors is added (equal to worm volume) to the tube and transferred to an ice-cold metal block; other sample buffers are also acceptable. Immediately after transfer, lysing of the worms is performed. An ice-chilled pestle (glass or plastic) is inserted into the bottom of the microfuge tube and rotated fifty to seventy five circular strokes until very little resistance is felt. At this point, the extract can be flash frozen and handled at a later time-point. For a smaller volume of extract, it is rather important to separate the extract from the sand to maximize the yield. This can be easily performed by filtering the sand extract through a small microfuge tube column with a small needle-size hole in its bottom. The hole should be covered with 100uL of clean glass beads (425-600 μm, Sigma G8772) and be placed on top of another empty clean microfuge tube. The stack of tubes can be assembled in a 15 ml blue cap tube. Once the sand extract is ready, the tube should be put on top the tube that has glass beads at its bottom. A clean injection needle can now be pushed through the bottom of the tube with sand extract until a hole is made. Centrifugation at 2K RPM for 1 to 2 minutes provides an extract that is virtually free from sand particles.
For RNA preparation, washed worms are harvested in Trizol solution (Life Technologies), at which stage they can be snap-frozen and stored at -80 0C for RNA extraction at a later time-point. Worms are ground in Trizol with sea sand and pestle as described above. After filtering of the sand, RNA is extracted using the PureLink RNA Mini Kit (Life Technologies) and eluted in RNAse-free ddH20. Extra caution should be taken to make sure that sand, tubes, solutions and tools are totally RNAse free. The sand should be baked at 240 °C for at least 4 hours and plastic RNAse-free pestles should be purchased.
*2X LDS sample buffer: 100 mM Na-Tricine pH 7.8, 100 mM DTT, 14% W/V Glycerol, 4% LDS, 0.05% CHAPS and 0.002% Bromphenol Blue
Acknowledgements: This work was supported by funding to BW from NIH (NS060872 and ES020395).
Figures
Articles submitted to the Worm Breeder’s Gazette should not be cited in bibliographies. Material contained here should be treated as personal communication and be cited as such and only with the consent of the author.
A modified leaving assay to study pathogen avoidance
C. elegans constantly encounter pathogenic threats and the ability to recognize and avoid these threats is crucial to survival. C. elegans have evolved a variety of immediate and learned behavioral responses to minimize pathogen exposure (Yook and Hodgkin, 2007; McMullan et al., 2012; and others). Behavioral responses to pathogens are commonly investigated using a food choice assay where animals are given a choice between their standard laboratory food, OP50, and the pathogen of interest (Yook and Hodgkin, 2007; McMullan et al. 2012). The results of this type of assay integrate both the known attractive effect of OP50 and the attractive/repellent effect of the pathogen under investigation. In order to examine only potential repellent cues, we have adapted the male leaving assay (Lipton et al., 2004) to analyze the response of naïve animals upon exposure to Microbacterium nematophilum, a gram positive bacteria which binds to and colonizes the rectal opening of C. elegans.
Method: 90 mm NGM assay plates were prepared using 13 ml NGM in order to make animals tracks easier to observe. Bacterial cultures were grown to OD600 = 1.0 before plating 20 µl in the center of the assay plate.
Single synchronized young adult hermaphrodites were transferred onto individual plates. 10 plates were prepared for each bacterial lawn to be tested.
Plates were kept at 20 oC and examined at 1 hr intervals to determine if the animal was a leaver, meaning that the animal or its tracks can been seen 1 cm or less from the edge of the plate. Animals determined to have left were not scored again at later time points
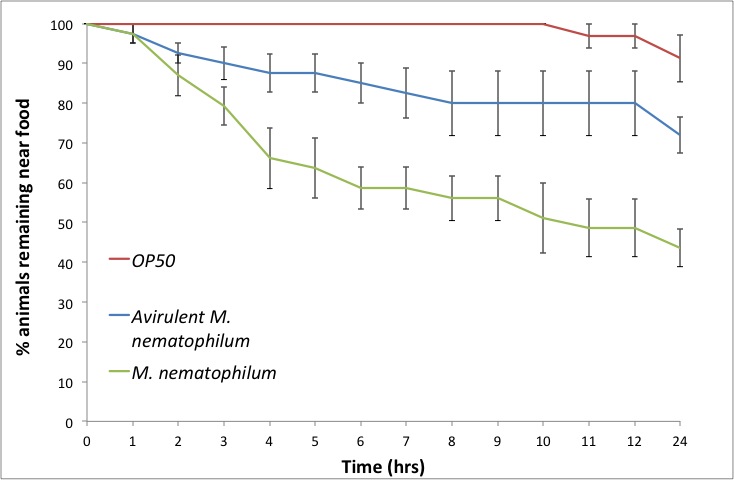
Results: Animals placed on plates seeded with OP50 show little leaving behaviour over 24 hrs. An avirulent M. nematophilum strain (Akimkina et al., 2006) induces a limited leaving response, whilst virulent M. nematophilum induces a rapid response over 24 hrs (Figure 1).
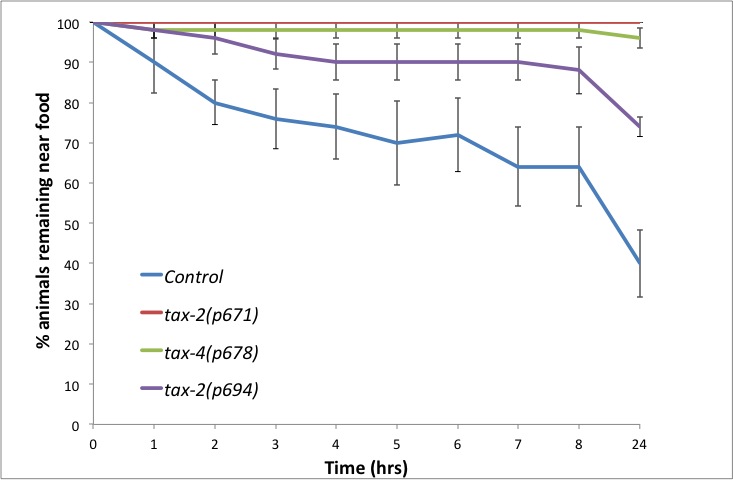
Using this assay we tested the role of the cyclic nucleotide gated ion channel TAX-2/TAX-4 in this behaviour. This pathway has been shown to be required for wild type responses to M. nematophilum using a food choice assay (Yook and Hodgkin 2007). Avoidance of M. nematophilum depends on TAX-2/TAX-4, since mutations in either TAX-2 or TAX-4 abrogate the leaving response (Figure 2). In wild-type animals TAX-2/TAX-4 is expressed in AWC, AFD, ASE, ASG, ASJ, AQR, BAG, ASK, ASI, AWB, and PQR (Coburn and Bargmann, 1996). The allele tax-2(p694) has a deletion in the promoter region and first exon of tax-2 abolishing its expression in ASE, AQR, AFD, and BAG (Coburn and Bargmann, 1996). In contrast to the response to M. nematophilum in a food choice assay (Yook and Hodgkin, 2007), tax-2(p694) animals show a defective leaving response particularly at longer time points (Figure 2), suggesting these four cells are important for the normal leaving response to this pathogen.
This simple behavioural assay could be adapted to investigate the response to numerous pathogens and removes the influence of the attractive cue from OP50 in food choice assays.
Acknowledgements: We thank Arantza Barrios (University College London) and Richard Poole (University College London) for suggestions and assistance in setting up this assay. We thank Jonathan Hodgkin for M. nematophilum strains.
Figures
References
Akimkina T, Yook K, Curnock S, and Hodgkin J. (2006). Genome characterization, analysis of virulence and transformation of Microbacterium nematophilum, a coryneform pathogen of the nematode Caenorhabditis elegans. FEMS Microbiol. Lett. 264, 145–151. 
Coburn CM, and Bargmann CI. (1996). A putative cyclic nucleotide-gated channel is required for sensory development and function in C. elegans. Neuron 17, 695–706. 
Lipton J, Kleemann G, Ghosh R, Lints R, and Emmons SW. (2004). Mate searching in Caenorhabditis elegans: a genetic model for sex drive in a simple invertebrate. J. Neurosci. 24, 7427-7434. 
McMullan R, Anderson A, and Nurrish S. (2012). Behavioral and immune responses to infection require Gαq-RhoA signaling in C. elegans. PLoS Pathog 8, e1002530. 
Yook, K. and Hodgkin J. (2007). Mos1 mutagenesis reveals a diversity of mechanisms affecting response of Caenorhabditis elegans to the bacterial pathogen Microbacterium nematophilum. Genetics 175, 681-697. 
L1 arrest affects dauer decision in C. elegans
During larval development C. elegans has two distinct time points where it can arrest development until environmental conditions become more favorable. The first one is L1 arrest, which occurs when worms hatch in the absence of food. The second decision point is later, around L1-L2 molt, and if conditions are poor at this time (little food, crowding, and high temperature) worms gain the potential to become dauers. L1 arrest and dauer formation are not mutually exclusive and, in principle, a worm can go through both of them sequentially. However, it is not known whether L1 arrest and dauer formation are coupled, i.e., if experiencing L1 arrest affects a worm’s decision to become a dauer. A naïve prediction would be that worms that experienced L1 starvation should be more susceptible to become dauers since they were already exposed to a harsh environment.
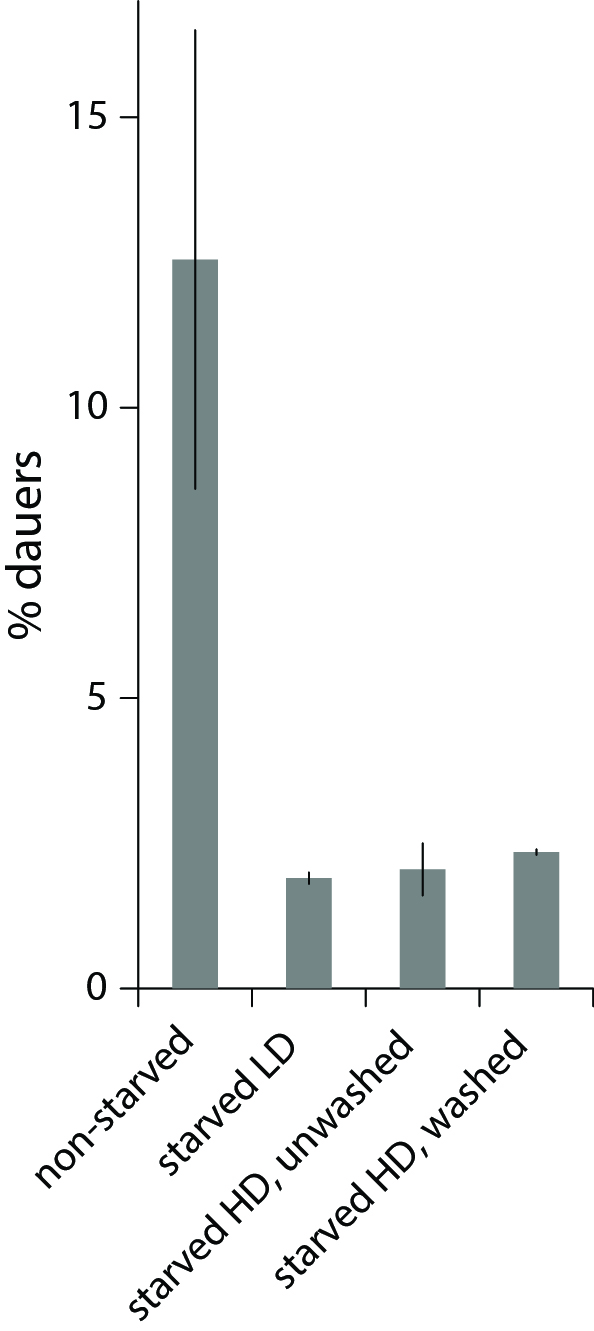
We studied dauer formation of N2 worms in liquid and found that in reality worms that experienced L1 arrest and starvation were less likely to become dauers. In a typical experiment, eggs obtained from an egg prep were incubated at 20 0C in S-complete with a small amount of bacterial food (HB101) and dauer formation was assessed 6 days later based on survival in 1% SDS (Pungaliya et al., 2009). When we let eggs hatch without food and added it 1-4 days later, keeping other conditions the same, significantly fewer worms became dauers (Figure 1). This was not due to slower development after starvation and incomplete dauer formation. We also found that this effect reflected an internal state of the worm rather than exposure to L1 exometabolome during L1 arrest since worms starved as L1s at low and high density later formed dauers with the same probability (Figure 1, during the dauer formation worms were at the same low density of 1 worm/µl to minimize the effect of released chemicals, see below).
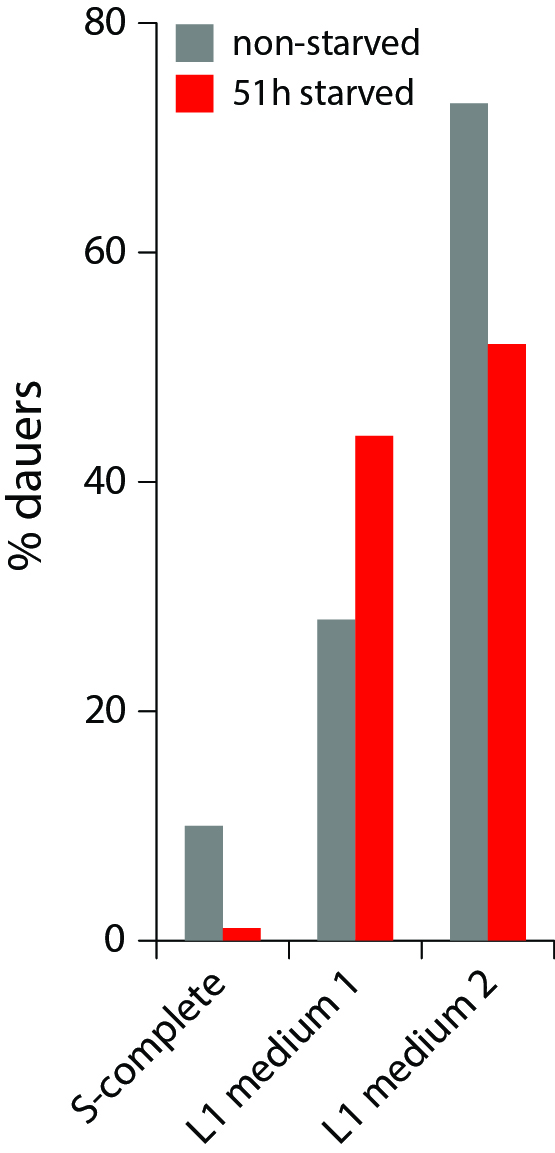
Starved L1 worms release numerous metabolites, including several ascarosides, and we anticipated that they might promote dauer formation. Indeed, when S-complete was substituted with conditioned medium from high-density L1 larvae starved for 24 h, more worms became dauers (Fig. 2), consistent with the known dauer-inducing effect of crowding mediated by ascarosides (Ludewig and Schroeder, 2013). However, this result apparently contradicts the starvation effect described above. On one hand, when a worm itself experienced L1 arrest but received no signal from other worms (low density condition), it was less likely to become a dauer and preferred to try to grow to adult. On the other hand, a worm that had not experienced starvation itself but was exposed to chemical signals released by other starved L1 worms, was more likely to become a dauer. The next obvious experiment is to combine these two opposite effects and see which one is stronger. The result of this test (Fig. 2) is that the external starvation signal (mediated by L1 conditioned medium) overrides the internal one (L1 arrest). When making a decision whether to become a dauer, apparently C. elegans worms trust the communal voice more than their own.
While the dauer-inducing effect of L1-conditioned medium is intuitively easy to accept, the negative effect of L1 starvation on subsequent dauer formation is less clear. A recently published model of contrast effects (McNamara et al., 2013) may provide a theoretical framework for understanding this phenomenon. But to keep us from being too comfortable with our simplistic views nature presented us with another puzzle: L1 arrest does not negatively affect dauer formation in C. briggsae.
Figures
References
Ludewig AH and Schroeder FC. (2013). Ascaroside signaling in C. elegans (January 18, 2013), WormBook, ed. The C. elegans Research Community. 
McNamara JM, Fawcett TW, and Houston AI. (2013). An adaptive response to uncertainty generates positive and negative contrast effects. Science 340, 1084-1086. 
Pungaliya C, Srinivasan J, Fox BW, Malik RU, Ludewig AH, Sternberg PW, and Schroeder FC. (2009). A shortcut to identifying small molecule signals that regulate behavior and development in Caenorhabditis elegans. Proc. Natl Acad. Sci. U. S. A. 106, 7708-7713.