The CRISPR/Cas9 system is rapidly becoming the genome-editing technique of choice in a variety of organisms (Pennisi, 2013; Frokjaer-Jensen, 2013). We recently applied the CRISPR/Cas9 module to target cis-regulatory elements in C. elegans.
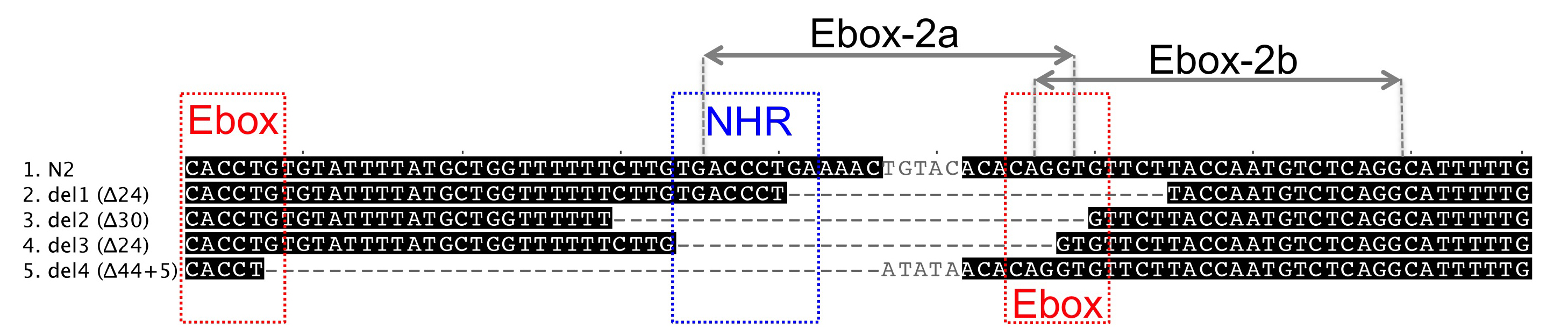
We focused on a cis-regulatory region that is necessary and sufficient to drive lin-3 expression in the anchor cell (Hwang and Sternberg, 2004). This region harbours three regulatory sites in close proximity to each other: one NHR binding site, which is required for anchor cell expression, and two surrounding E-boxes on complementary DNA strands (Hwang and Sternberg, 2004). Interfering with these elements is expected to decrease lin-3 expression in the anchor cell and therefore lead to vulval hypo-induction as a phenotypic consequence.
Among current available methods, DNA-based delivery of the CRISPR/Cas9 complex has been reported to give rise to small deletions, like those presumably required to target specific cis-regulatory elements (Frokjaer-Jensen, 2013). We therefore used the plasmid vectors and protocol described in the first DNA-based CRISPR report in C. elegans (Friedland et al., 2013). To generate a pU6::lin-3 single guide RNA, named Ebox-2a, we used fusion PCR, as previously described (Friedland et al., 2013). However, instead of cloning first the PCR product into a vector, we PCR-purified and added it directly to the injection mix. We injected 15 N2 animals with a mix consisting of 40 ng/μl of eft-3::cas9 plasmid, 40 ng/μl of pU6::lin-3 sgRNA PCR product, 10 ng/μl of myo-2::GFP as co-injection marker and 10 ng/μl of pBluescript plasmid to reach a final 100 ng/μl concentration.
We recovered 4 injected animals segregating GFP-positive F1 progeny. From these 4 plates, we singled 29 F1 animals for further characterisation. We noticed that some transgenic F1 animals already showed a striking egg-laying defective (Egl) phenotype, which is the phenotype expected upon lin-3 reduction-of-function. However, when we sequenced Egl F1s, we found no deletion in the region of interest and the Egl phenotype was not heritable in the F2 generation. We conclude that, similar to other systems, Cas9 alone is sufficient in C. elegans to antagonise specific transcription factors that mediate transcription (Pennisi, 2013).
The progeny of some F1 animals isolated from 3 out of 4 P0 plates continued expressing GFP in the F2 generation and segregated back the Egl phenotype. We singled Egl worms and looked for heritable transmission in the subsequent generations. We identified four cases where the Egl phenotype was stable in the progeny and identified in all cases small deletions removing partly or entirely the NHR binding site and one E-box. We then sequenced the F1s and found that two deletions were already present in the F1 generation, whereas the remaining two occurred during the following generations.
We also designed another pU6::lin-3 sgRNA, Ebox-2b, targeting the nearest possible site to the previous construct, as defined by the G/A(N)19NGG rule. This construct was injected as a plasmid at 150 ng/μl concentration (300 ng/μl final concentration in the injection mix). We recovered 4 out of 15 injected animals segregating GFP-positive F1 progeny and singled 12 F1 animals for further analysis. Despite a frequent Egl phenotype in the F2, we found no heritable transmission of the phenotype and no molecular deletion in the F1s.
From the above experiments we draw the following conclusions:
1. sgRNAs can be injected as purified PCR products in C. elegans. This further simplifies the generation of targeting constructs for DNA-based genome editing, as it eliminates the single cloning step in the protocol.
2. When targeting cis-regulatory elements by CRISPR, the phenotypes of interest should be tested for their heritability.
3. We found evidence that CRISPR-mediated deletions can occur not only in the F1, but also in subsequent generations.
4. The efficiency of generating deletions can vary even when targeting directly adjacent genomic regions. Therefore, multiple sgRNAs should be tested for successful genome editing.
Figures
References
FriedlandAE, Tzur YB, Esvelt KM, Colaiacovo, MP, Church GM, and Calarco, JA. (2013). Heritable genome editing in C. elegans via a CRISPR-Cas9 system. Nat. Methods 10, 741-743. 
Frokjaer-Jensen, C. (2013). Exciting prospects for precise engineering of Caenorhabditis elegans genomes with CRISPR/Cas9. Genetics 195, 635-642. 
Hwang BJ, and Sternberg PW. (2004). A cell-specific enhancer that specifies lin-3 expression in the C. elegans anchor cell for vulval development. Development 131, 143-151. 