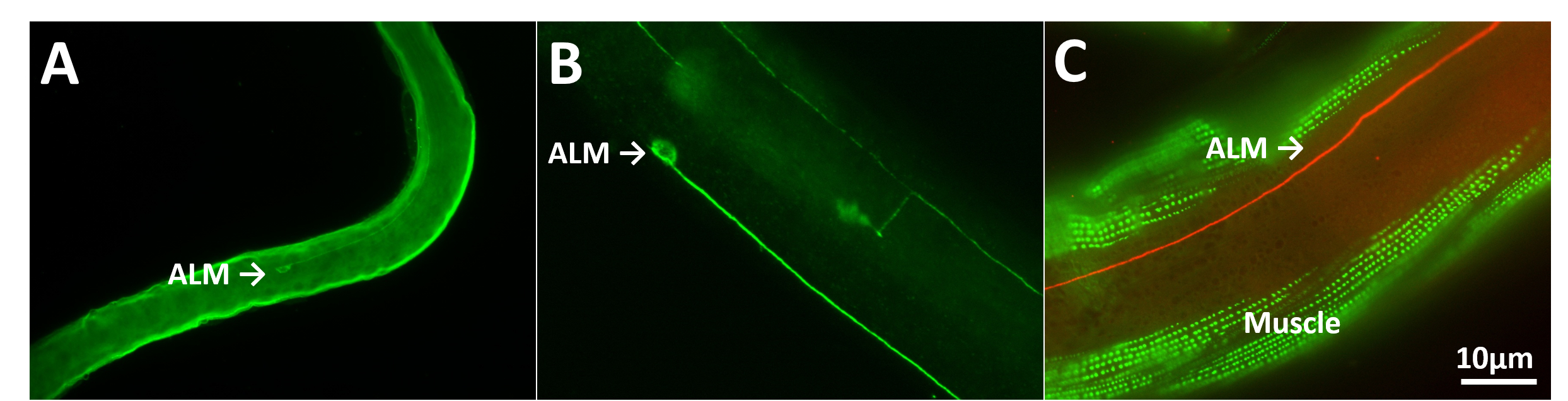
To compare antibody staining over time, one needs to have a method of archiving tissue samples. In general, however, C. elegans prepared for histology have not been archived for long periods. For example, worms mounted on slides sealed with nail polish and stored at 4°C (Wilson, et al., 2012; Duerr, 2013) last two weeks to a month (Duerr, 2013). After this time, significant tissue shrinkage occurs and fluorescent signals rapidly decay (Figure 1A). I have found that the polyvinyl pyrrolidone-based cryoprotectant known as “anti-freeze” (Tebbenkamp, et al., 2011) that is used to archive partially and fully processed mammalian tissues for long periods works well with histologically prepared C. elegans.
I prepared animals using the staining method of Finney and Ruvkun (Finney and Ruvkun, 1990) with antibodies against MEC-18 and MEC-2 or the 4′,6-diamidino-2-phenylindole (DAPI) probe against DNA or the actin-binding reagent phalloidin. Animals were then archived with or without anti-freeze for varying times. In contrast to sealing with nail polish no significant degradation of tissue morphology or fluorescence was seen for at least a year (Figure 1B-C). Similar results were observed with worm cell cultures. Thus, anti-freeze did not disrupt antibodies bound to their epitopes. In addition, animals expressing GFP and mCherry that were observed after several months in anti-freeze remained intensely fluorescent as were animals stained following long-term storage in anti-freeze. In general, the morphology of touch receptor neurons and other cells was indistinguishable in worms that were archived in anti-freeze compared to worms that were immediately analyzed. Moreover, morphology remained intact even after several freeze/thaw cycles.
To archive, mix 50 ml worm pellet and 500 ml anti-freeze (100 mM sodium acetate, 250 mM polyvinyl pyrrolidone (PVP), and 40% ethylene glycol, pH 6.5), and store at -20°C. The anti-freeze itself can be stored for years at 4°C or -20°C.
To recover worms for histology, pellet animals and wash 2-3 X with M9 buffer.
Figures
References
Duerr JS. (2013). Antibody staining in C. elegans using “freeze-cracking”. J. Vis. Exp. 80, doi: 10.3791/50664. 
Finney M and Ruvkun G. (1990). The unc-86 gene product couples cell lineage and cell identity in C. elegans. Cell 63, 895-905. 
Tebbenkamp AT, Green C, Xu G, Denovan-Wright EM, Rising AC, Fromholt SE, Brown HH, Swing D, Mandel RJ, Tessarollo L, and Borchelt DR. (2011). Transgenic mice expressing caspase-6-derived N-terminal fragments of mutant huntingtin develop neurologic abnormalities with predominant cytoplasmic inclusion pathology composed largely of a smaller proteolytic derivative. Hum. Mol. Genet. 20, 2770-2782. 
Wilson KJ, Qadota H, and Benian GM. (2012). Immunofluorescent localization of proteins in Caenorhabditis elegans muscle. Methods Mol. Biol. 798, 171-181. 