 Table 1: Primers for amplifying the pPK719 cassette; pCC1FOS homology arms are shown in brackets. Note that the pCC1Fos homology sequence in PK951 is also present in the pBSKSII(+) vector backbone.
Table 1: Primers for amplifying the pPK719 cassette; pCC1FOS homology arms are shown in brackets. Note that the pCC1Fos homology sequence in PK951 is also present in the pBSKSII(+) vector backbone.
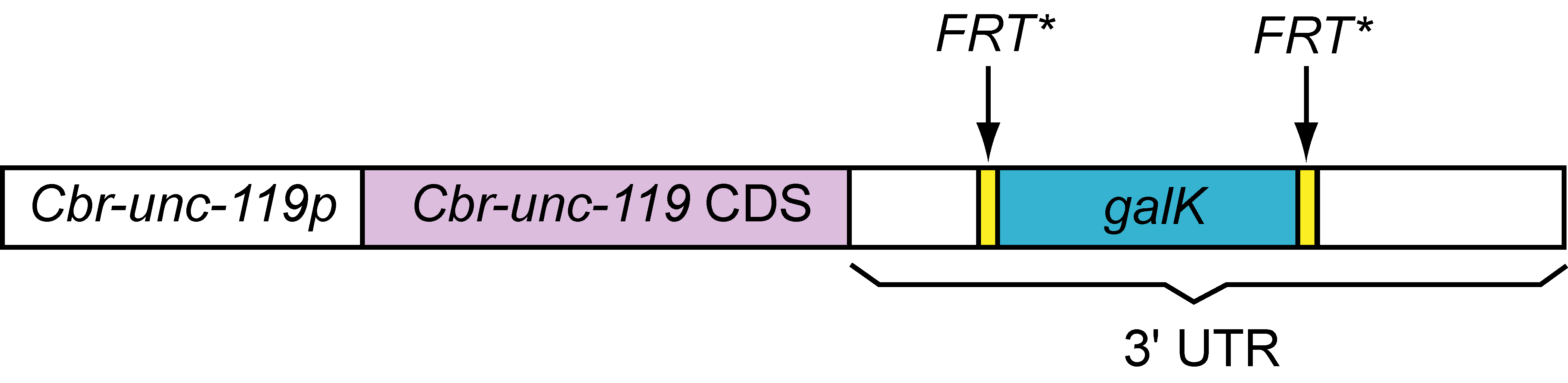
The 3.6 kb Cbr-unc-119::FRT*::galk::FRT* cassette, which is PCR amplified from pPK719 using the PK951/PK952 primer pair (Table 1), can be targeted by recombineering to insert between nts 281:282 in the pCC1FOS fosmid vector backbone. In other words, this cassette can be readily inserted in all fosmids contained in the C. elegans library developed by Don Moerman and colleagues. We have tested the construct in fosmid recombineering and have shown that it rescues unc-119(ed3) mutants. The clone will be made available at http://www.Addgene.com.
Figures
References
Maduro M, and Pilgrim D. (1996). Conservation of function and expression of unc-119 from two Caenorhabditis species despite divergence of non-coding DNA. Gene 183, 77-85. 
Praitis V, Casey E, Collar D, and Austin J. (2001). Creation of low-copy integrated transgenic lines in Caenorhabditis elegans. Genetics 157, 1217-26. 
Tursun B, Cochella L, Carrera I, and Hobert O. (2009). A toolkit and robust pipeline for generation of fosmid-based reporter genes in C. elegans. PLoS One 4, e4625. 