Primary cell culture techniques for embryonic and larval cells of C. elegans are well established (Christensen et al., 2002; Zhang et al., 2011). These cell culture methods utilize L-15 complete medium, which is prepared by the addition of FBS (fetal bovine serum) and sucrose to commercially available L-15 medium. L-15 medium is versatile as it does not include carbonate and hence does not require a CO2 incubator to maintain the optimal pH. Although L-15 complete medium is sufficient for the maintenance and differentiation of muscle cells and neurons in primary culture, there is little published information on the effects of varying culture conditions. Therefore, it seemed worthwhile to examine the comparative performance of other culture media.
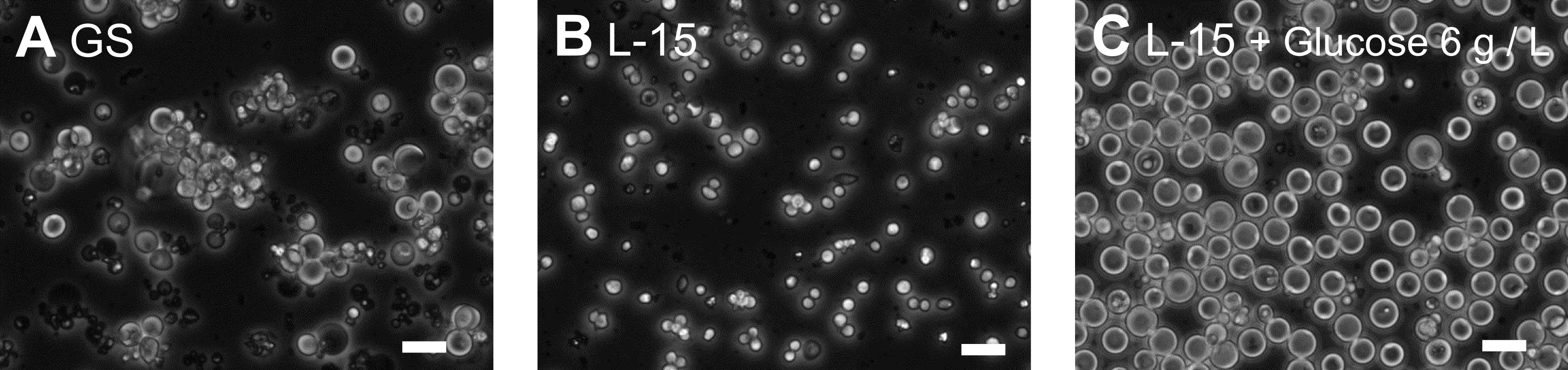
We isolated primary cells from embryos using the established method (Christensen et al., 2002) and compared the growth of the cells in a number of commercially available culture media to which we added FBS. By comparison to other media, we found that GS medium, which is formulated for use in culture of mouse germline stem cells (Kanatsu-Shinohara et al., 2003), supported enhanced growth of the C. elegans cells (Figure 1A, B). Examination of the ingredients of GS medium suggested that its relatively high concentration of D-glucose was likely the principal factor for this growth enhancement effect. To determine whether this was the case, we added 6 g/L of D-glucose to L-15 complete medium and found enhanced cell growth. In medium with D-glucose, the diameter of the cells averaged more than 10 mm after two weeks of culture, while in standard medium they averaged about 5 mm (Figure 1B-E).
Typically, the cultures were initiated using 106 cells in 1.5 ml medium in 25 ml plastic culture flasks and incubated at 20°C. The addition of 6 g/L D-glucose caused about 10% of the embryonic cells to swell. The enlarged cells adhered weakly to the bottom of uncoated culture vessels and tended to form cell clumps. We cultured the embryonic cells using weekly addition of 500 ml fresh medium and a monthly change of the medium by collecting cells through centrifugation (1000 rpm, 3 min). With this periodic medium change, some of the enlarged cells survived more than 3 months.
Although, the cell population appeared homogeneous, examination of cultured cells from a transgenic strain with the chromosome integrated transgene Is[myo-2p::venus, rab-3p::NLS::mRFP] revealed the presence of several cell types. The fluorescent reporter genes Venus and mRFP, which were expressed under the control of the myo-2 and rab-3 promoters, respectively, showed at least 3 cell types were present: Venus+, mRFP+, and Venus-mRFP-. Expression of the Venus reporter gene was seen in only some of the enlarged cells, while that of mRFP was only present in small cells (Figure 2A-C). The cells showed BrdU incorporation only on the first day of dissemination indicating that they did not proliferate after enlarging (data not shown). Although the cell culture is not permanent, we expect that this method will provide cells that can be applied to a range of investigations.
Figures

References
Christensen M, Estevez A, Yin X, Fox R, Morrison R, McDonnell M, Gleason C, Miller DM 3rd, and Strange K. (2002). A primary culture system for functional analysis of C. elegans neurons and muscle cells. Neuron 33, 503-514. 
Kanatsu-Shinohara M, Ogonuki N, Inoue K, Miki H, Ogura A, Toyokuni S, and Shinohara T. (2003). Long-term proliferation in culture and germline transmission of mouse male germline stem cells. Biol. Reprod. 66, 612-616. 
Zhang S, Banerjee D, and Kuhn JR. (2011). Isolation and culture of larval cells from C. elegans. PLoS One 6, e19505. 
Articles submitted to the Worm Breeder's Gazette should not be cited in bibliographies. Material contained here should be treated as personal communication and cited as such only with the consent of the author.
Leave a Reply
You must be logged in to post a comment.