Worm Breeder's Gazette 9(3): 96
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
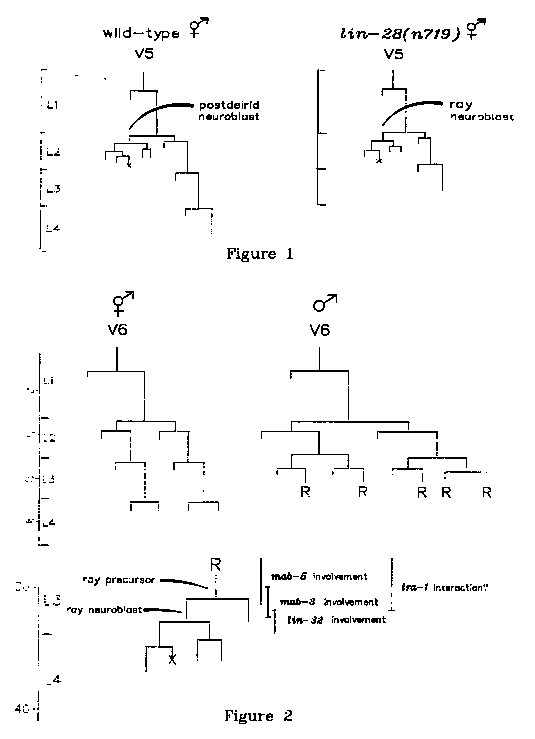
The precocious heterochronic mutation lin-28(n719) possesses an
unexpected phenotype: in hermaphrodites and males, an ectopic ray
neuroblast lineage is frequently found in place of a postdeirid (
figure l; Ambros and Horvitz, Science 226, 409). How can a ray, a male-
specific structure, form in the temporally transformed lin-28
hermaphrodite? Kenyon and Hedgecock (WBG 8 #2, 19) have proposed that
the fate of neuroblasts is influenced by the developmental stage in
which they arise: the neuroblast in the postdeirid position is
normally formed in L2, and therefore follows a postdeirid fate, but
the equivalent lin-28 neuroblast finds itself in a later temporal
environment, becoming a ray instead. Consistent with this hypothesis,
I have found that the ectopic ray does not result from a general
sexual transformation in lin-28 hermaphrodites, because it can still
form in lin-28(n719);tra-1dom(e1575)/+ XX and XO females (4/12 sides
examined by DAPI staining had the ectopic ray).
The lin-28 ectopic ray can be used to test whether mab-5 or mab-3,
two genes involved in male development, are required for the
expression of a ray neuroblast lineage. The null mutation mab-5(e1239)
results in ray precursor cells failing to form in the V5 and V6
lineages (Kenyon, Cell 46, 477); the null mutation mab-3(e1240) causes
ray precursor sublineages to produce seam cells instead of rays (at
least for V5 and V6 descendants; WBG 9 #2, 62). Compared with lin-28
alone (33/82 sides examined = 40%), the frequency of ectopic ray
formation is not significantly altered in mab-5;lin-28 (22/68 = 32%)
or in mab-3;lin-28 (19/67 = 28%) hermaphrodites or males, as assayed
by DAPI staining. It is therefore likely that the mab-5 and mab-3 gene
products are not necessary for a ray neuroblast lineage (unless the
lin-28 ectopic ray is abnormal, or behaves as a T-derived ray). In
contrast, the gene lin-32 does appear to be required for both wild-
type and lin-28 ray neuroblast lineages (0/15 lin-28;lin-32 sides had
ectopic rays; Kenyon and Hedgecock, ibid; C. Kenyon, personal
communication).
mab-5, s each affect a
different step in the sequence of divisions specific to the male
lateral hypodermis, all three causing seam cells to be made instead of
rays. It is interesting to speculate that their gene products may be
involved in a series of decisions which switches these lineages from a
hermaphroditic seam cell 'ground state' to the specialized male ray
sublineages (figure 2). In addition, tra-1 might interact negatively
with mab-5, and perhaps mab-3, but not lin-32, to control these
sexually dimorphic decisions (based on epistasis experiments). In this
sequential switch model, mab-5 and mab-3 products are not directly
involved in specifying a ray neuroblast lineage, consistent with the
observations above. This reasoning supports the neuroblast fate
hypothesis of Kenyon and Hedgecock in explaining how the lin-28
ectopic ray could arise in the presence of tra-1 activity, and argues
against a role for lin-28 in the generation of sexual dimorphism, as
has been suggested (Hodgkin, JEEM 83 Supp., 103). Furthermore, these
speculations provide a framework for understanding how the selector
gene tra-1 might control 'downstream' functions to implement sexually
dimorphic decisions at the level of individual cell fates.
{Figure 1}