Worm Breeder's Gazette 9(2): 95
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
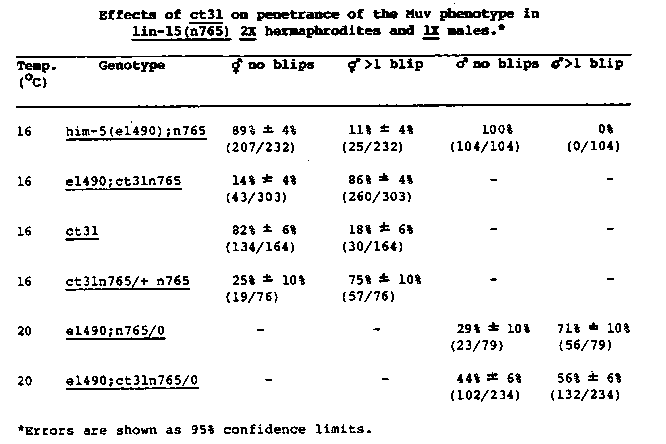
We have been analyzing an X-linked dominant suppressor of the semidominant allele her-1(n695)V, which causes incomplete masculinization of XX animals (Trent et al., 1983, Genetics 104:619). The suppressor, called ct31, was isolated from the F1 progeny of gamma-ray mutagenized n695 worms. Two- and three-factor mapping data show that ct31 is closely linked to unc-9 X. So far at least three phenotypes of ct31 have been identified. First, this mutation dominantly suppresses the Tra and Egl phenotypes of n695. Second, ct31 homozygotes are Dpy. ct31/+ heterozygotes are of variable length ranging from that of the homozygous Dpy to wild type. ct31 males are short and have abnormal tail structures. Third, recent evidence suggests that ct31 dominantly lowers X-chromosome expression in hermaphrodites but not in males. To investigate the effect of ct31 on X-chromosome expression, strains were constructed that carry ct31 and an X-linked hypomorph, lin- 15(n765). Mutations in lin-15 result in a Muv phenotype with one to six ventral protrusions (blips) (Ferguson and Horvitz, 1985, Genetics 110: 17). The penetrance of this allele varies at different temperatures as well as with the level of X- chromosome expression (P. Meneely, E. Ferguson, and W.B. Wood, manuscript in preparation). A mutation causing an increase in X- chromosome expression in a lin-15(n765) background will suppress the Muv phenotype (decrease the penetrance), while a mutation causing a decrease in X-chromosome expression will enhance the Muv phenotype ( increase the penetrance). The data in Table 1 show that ct31 enhances the penetrance of the lin-15(n765) Muv phenotype in hermaphrodites but not in males. An increase in penetrance is also seen in ct31/+;n765 hermaphrodites. One interpretation of these data is that ct31 lowers X-chromosome expression. Strains carrying ct31 and other X linked hypomorphs will be constructed to test this interpretation. We have independently isolated two other X-linked n695 suppressors, which result in phenotypes similar to those seen with ct31. Since ct31 males will not mate, complementation tests have not been done to ask if these three mutations are allelic. Over an eight-month period at least five strains carrying ct31 and two strains carrying one of the other Dpy suppressors have spontaneously reverted to wild-type length. The ct31 mutation could not be recovered from the ct31 revertants, suggesting that they did not result from an extragenic event. The X chromosome from a revertant strain acted as a wild-type chromosome when made heterozygous with ct31. Two of these revertants tested no longer suppressed her- 1(n695) or affected the penetrance of lin-15(n765) at 16 C. An attempt to isolate EMS induced alleles of ct31 was unsuccessful after screening 13,000 F1 progeny of her-1(n695). Since ct31 was isolated after gamma-ray mutagenesis and spontaneously reverts to wild-type phenotype at a high frequency, it may be an unstable chromosomal rearrangement. The dominant ct31 phenotype contrasts strikingly with the recessive phenotype resulting from egl- 16 (n485) (Anne Villeneuve and Barbara Meyer, WBG, Vol. 9, no. 9). ct31 has a feminizing effect and seems to lower X-chromosome expression while egl-16(n485) has a masculinizing effect and increases X-chromosome expression. Differences in map position rule out the possibility that the two mutations could be allelic (Anne Villeneuve and Barbara Meyer, personal communication). Therefore, ct31 may represent one of several loci on the X chromosome involved in interpreting the X-chromosome-to-autosome ratio in the control of sex determination and dosage compensation. [See Figure 1]