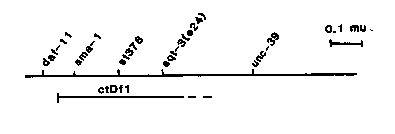Worm Breeder's Gazette 9(2): 30
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
The gene myo-3 which codes for the minor body wall myosin heavy chain myoA (Miller et al PNAS in press), has been localized to LGV near Sma-1 (Albertson, EMBO 4, 2493-2498). Increased accumulation of myoA through tandem duplication of the gene suppresses unc-54(0) mutations, but hypomorphic or null alleles of the gene have not been identified. One postulate has been that myo-3(0) mutations when homozygous are lethal. To look for lethal mutations that might lie in the myo-3 gene, Kathy Kellerman, a rotation student at Washington University, and I screened over 700 mutagenized F1 clones of a sma- 1/daf-11 strain (see map below) for clones failing to produce expected numbers of normal Sma-1 progeny. Of the several mutations identified, one, st378, resulted In the arrest of homozygous animals as paralyzed L1's. The st378 mutation has several properties consistent with the hypothesis that it is a myo-3 hypomorphic allele as described below, but the relatively small numbers of purified homozygotes obtained so far have frustrated efforts to measure myoA levels directly. The st378 mutation is closely linked to, and right of, Sma-1 as shown on the map below, based on 2 and 3 factor data. Although the Sma-1 mutation has been separated from st378, a chromosome carrying only gt378 has not been obtained yet. The Sma-1(e30) st378 homozygotes proceed normally through embryonic cell divisions and resemble Sma-1 in morphogenesis, with the exception that the double mutant never changes its position achieved at the end of elongation. Only slight movements of the tip of the head and tail are visible. Hatching occurs only slightly later than the wild type sibs. The pharynx pumps and bacterial ghosts are present in the L1 gut, yet the animal arrests in the L1 stage without ever unfolding. ctDf1/e30 st378 animals have a similar if not identical phenotype. Electron microscopy of Sma-1(e30) st378 homozygotes show that the body wall myofilament lattice is abnormal. Few normal thick filaments are present; in addition there are filaments larger than the normal thick filaments of irregular outline. The st378 is essentially recessive in a wild type background, but is semi dominant in unc-56(e190) or unc-15(e73) backgrounds. The unc-54( e190); ) st378/++ animals for example are more severely paraiyzed than unc-54(e190) homozygotes, and the few animals of this genotype that reproduce have very small brood sizes. sup-3 mutations in trans counterbalance these effects; e.g. unc-54(e190); 07)/sma-1(e30) st378 animals resemble unc-54(e190) homozygotes, and are more paralyzed than unc-54(e190); 07)t+ animals. The ability in the nematode to identify a protein of interest, to recover its DNA and to localize it by cytogenetics, and to obtain subsequently mutations in the genomic copy of that gene is an approach of considerable potential.