Worm Breeder's Gazette 9(2): 13
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
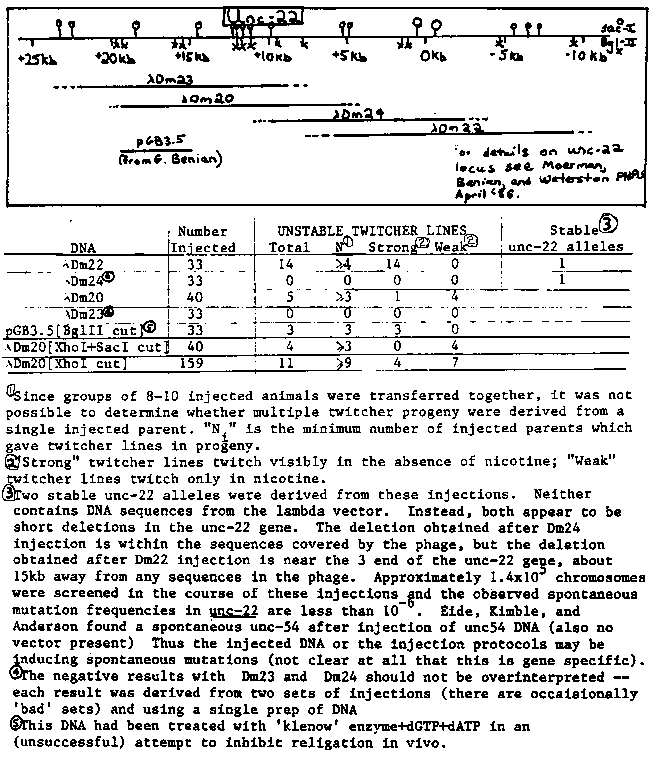
Twitching behavior provides a very sensitive assay for differences in unc-22 activity levels. Worms with very low unc-22 activity twitch under normal conditions, while worms with less drastic reductions in unc-22 activity can be induced to twitch by placing them in solutions of one percent nicotine (see table:entries reflect polyploid phenotypes; D.M. unpublished). [See Figure 1] In particular, the behavior of heterozygous mutants in nicotine has been extremely useful to date in allowing facile selection of unc-22 mutants induced by a variety of agents. We hoped to use nicotine to select for homologous disruption of the unc-22 gene after injecting cloned pieces of the unc-22 gene. Although no such homologous disruption was observed, we have stumbled on a very efficient method for selection and maintenance of transformed lines with extrachromosomal tandem arrays. Various recombinant lambda and plasmid DNAs were injected into hermaphrodite gonads, and the next generation screened for twitching in 1% nicotine. With several of the DNAs, such animals were obtained at unexpectedly high frequency. When the progeny of these animals were followed, the twitching phenotype was found to be heritable, but no true breeding 'homozygote' strains could be obtained. A similarity to the extrachromosomal tandem arrays originally described by Stinchcomb, Shaw, Carr, and Hirsh (MCB December '85) was suggested by the presence of the injected DNA at very high copy number, and by the presence of extrachromosomal free duplication like fragments in mitotic and meiotic chromosome preparations (we are grateful to Donna Albertson for making and analyzing the stained chromosome preparations!!). The twitching phenotype correlated with the presence of the injected DNA and with the presence of the novel chromosome fragments. Thus extrachromosomal tandem arrays of unc-22 containing DNAs somehow interfere with unc-22 function. We do not yet know why this is true (sorry). The four major possibilities are I. Aberrant protein products could be produced which interfere with muscle function to cause twitching. II. Regulatory sites contained within the injected DNAs could compete with the endogenous unc-22 gene for specific factors required for expression. III. The extrachromosomal tandem arrays could be transcribed somewhat indiscriminately, leading to the production of 'antisense' RNA. The aberrant transcripts could conceivable come from minor promoters either in unc-22 or in vector sequences, and the amount and activity of the antisense RNA would be expected to depend critically on the exact structure of the template. IV. (far out) Some type of synapsis between the extracromosomal array and the endogenous unc-22 gene might occur in muscle cells, thereby disrupting expression. We cannot as yet rule out any of these possibilities, but the ability to disrupt activity using two recombinants with no overlapping sequences (lambdaDm22 and pGB3.5) makes explanations I and II slightly less plausible (although one should note that protein motifs and/or cis acting regulatory sequences could easily be repeated throughout the gene.) In any case, this selection provides the most rapid procedure to date for creating transformed worm lines. As noted in the table below, different unc-22 recombinants give different spectra of phenotypes among the transformed progeny. With phage lambdaDm22, 14 strong twitcher lines were obtained (twitching observed in the absence of nicotine) among the progeny of 33 injected worms. Including total time required for injection, recovery, scoring and following of progeny this works out to approximately 30 minutes per transformed line. Conceivably your favorite gene or segment could be incorporated into the tandem arrays either by linking to unc-22 clones, injecting and selecting in nicotine, or by coinjecting your fragment cloned into the same vector as the unc-22 segment [worms are able to make mixed tandem arrays of coinjected homologous plasmids (R. Jefferson, personal communication)]. Of course there are a few caveats: it's not clear yet exactly what features of the injected recombinant are crucial for the twitcher phenotype and it's not known whether tandem arrays are compatible with properly regulated expression (particularly mixed arrays with unc-22). Note that many experiments (such as the introduction of putative regulatory sequences at high copy number to compete out specific effectors) may not require proper expression of the reintroduced DNA. The recombinants lambdaDm22 and pGB3.5 are freely available. Have fun. [See Figure 2] INJECTIONS: Both N2 and rec-1(s180) [Rose and Baillie Nature 281, 599] animals were used for injection. No consistent difference was found between the two strains. Animals were injected as young adults, 5-10 times in both gonads. The injected animals had been reared at 20 C, but were shifted up to 25 C immediately after injection. After 18- 24 hours, the animals were transferred so that each set of injections yielded an 'early' brood and a 'late' brood. Between 75 and 90% of the animals were fertile after injection (as judged by the presence of young eggs in the proximal arm of the gonad after 18-24 hr). SELECTIONS: F1 progeny (as L4s and young adults) were rinsed off plates in 1% nicotine (in water) and scored for twitching after 5-15 minutes at room temperature. After this the wild type animals start to wake up. Contaminated or starved plates tend to give a background of fakes. Most (but not all) of the tandem array containing strains appeared in the 'early' brood. A number of animals which clearly twitched in nicotine but which did not transmit the trait to any of their progeny were observed (these may be F1 mosaics). SEGREGATION: For each line, the strongest twitching animals were picked for several generations (in nicotine if necessary). In order to confirm the instability, animals which did not twitch in nicotine were cloned and the absence of twitching progeny confirmed in nicotine. A small fraction of the non-twitching animals always give twitching progeny (mosaicism?, noise?). Different subcloned strains appear to have different transmission frequencies (up to 95%) and this may correlate within a line with the copy number of free dups and with the severity of phenotype. Many of the lines exhibit a weak him phenotype with frequent twitching of XO progeny (dup disjoins from X?). BLOTS: The high copy numbers of the extrachromosomal arrays make the DNA easy to pick up on blots. This has been done for representitive strains. It is convenient to pick individual twitching animals from each strain, confirm twitchers among the progeny, and harvest the F2 generation from each plate (one plate gives enough DNA for many blots). Precise copy number estimates are a bit difficult to estimate because DNA was prepared from mixed populations; For the lambda phage recombinants, between 40 and 200 copies are present in the derived tandem arrays. The strains injected with pGB3.5/BglII/ have a distinct band at 2.7kb (not present in N2 DNA) after a SacI digest of genomic DNA is stained with EtBr. This band is apparently the puc vector and is strongest in a line where strong twitchers have been continually selected.