Worm Breeder's Gazette 9(2): 11
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
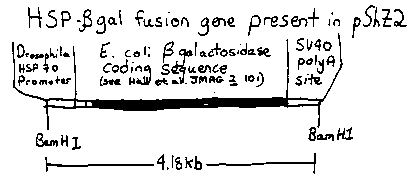
In the last gazette, I reported that coinjection of one plasmid carrying the amber suppressor sup-7 (used as a selectable marker) with a second, unselected plasmid could be used to obtain stably transformed lines which contain sequences from the second plasmid in addition to sup-7 DNA. I have used this to introduce a plasmid pShZ2 ( from Sean Munro) carrying a Drosophila heat shock promoter (HSP) fused to sequences coding for E. Coli -galactosidase (see diagram). Of three independent stably transformed lines derived from these injections, two (CB4027 and CB4028) were analyzed on southern blots and found to contain the HSP- -gal fusion segment in apparently unrearranged form. -galactosidase activities from these transformed lines have been compared with control lines both by soluble enzyme assays and by histochemical staining. 1. SOLUBLE ENZYME ACTIVITIES: Worms were grown on a bacterial strain deleted for the -galactosidase gene , harvested as L4s and homogenized by sonication (painful on the ears) or by a new 'sandblasting' method (see below). The resulting lysates were incubated with ONP -Galactoside (yellow product) or 4- Methylumbelliferyl -Galactoside (fluorescent product) and enzyme activities determined spectrophotometrically. A very low basal level of enzyme activity was detected both in the parental line, and in sup- 7 transformed lines not carrying the HSP- -gal fusion. This basal level was not affected by heat shock (3 hours at 34 C). Under normal growth conditions (20 C) the two lines carrying the HSP- -gal fusion produce levels of -galactosidase similar to the parent line (within 2- fold). After heat shock, a striking increase of 10-20 fold in galactosidase level is seen with both CB4027 and CB4028. 2. HISTOCHEMICAL STAINING: Whole worms were fixed and stained for gal activity as described below. No staining was observed in the parental line (with or without heat shock) or in CB4027 or CB4028 growing at 20 C. After heat shock, strong and reproducible staining of both CB4027 and CB4028 was observed (>95% of the animals stain). The staining appears almost exclusively in the pharynx (all stages) and in embryos. The localization of the stain is unlikely to reflect a simple permeability difference, since squashed and/or cut animals show similar patterns of staining. Additionally, injection of pure enzyme into the gonad, gut or pharynx of wild type animals followed by fixation and staining as above resulted in specific staining in each case of only the injected tissue. Histochemical staining is being used to examine the segregation of the inducible galactosidase activity: Preliminary evidence suggests that multiple loci (2-3) in CB4027 carry functional HSP- -gal segments. In particular at least one locus unlinked to the amber suppression activity (which maps on chromosome IV(L) near unc-17) can confer heat inducible gal activity. This is consistent with estimates of copy number for the gene fusion, between 5 and 20 for the two strains. The possibility of extrachromosomal inheritance for the transgenic HSP- - gal loci cannot yet be ruled out but seems unlikely due to the DNA copy number and the fact that inducible gal activities have been maintained in the two strains in the absence of selection for over 30 generations. A rough estimate of the induced -Galactosidase as a fraction of total protein can be obtained either from soluble enzyme assays or from histochemistry (by comparison with injections of pure enzyme). These estimates on the order of .001% and .01% respectively (the difference may reflect losses in solubilizing the enzyme). Why is staining in induced animals limited to the pharynx? Obvious suspects include the nature of the worm heat shock response, the synthetic rates and stabilities of -gal protein and message in different tissues, and possible fortuitous expression signals in the HSP- -gal fusion plasmid that was used. In any case the strains may prove useful in finding nematodes on warm summer nights. SAND EXTRACT: To worms (10-100 l) in a 1.5ml microfuge tube are added about equal volumes of buffer and acid washed fine grain sand. The tube is then agitated vigourously-- I do this by striking the bottom of the tube along the pegs of a plastic (Gilson) test tube rack 10-20 times (like bad, loud guitar playing). Sand can be removed by centrifugation or filtration. All the worms are lysed and completely broken up by this procedure and recovery of total protein and enzyme activity appear comparable to sonication. -GAL STAIN: Wash worms in water and place in drop on a slide (I use 4-well multitest slides). Dry slides 5-10 min in a dessicator jar under vacuum (<2mbar). Dip the dried slides in acetone and dry on benchtop. Add 100 l of staining solution [0.2M NaPhosphate pH 7.5, 1. 0mMDTT; saturate with substrate (6Bromo2NapthylBDgalactoside from Sigma) and add 0.004% SDS] for 1 hr at room temperature. Then add 20 l of 0.125% 'fast blue B diazonium salt' [BDH], mix well, and staining should appear within 10-20 min. The substrate is cleaved by - galactosidase to yield a local insoluble precipitate of 6Bromo2Napthol; this reacts with the diazotized Fast Blue to form insoluble turquoise dye (Rutenberg et al J. Histochem. and Cytochem. 6, 122). The fast blue must be dissolved (H20) just prior to use or a nasty orange stain develops which can obscure the reaction product. Some staining near the mouth in induced live worms can be obtained using this protocol without drying and acetone steps. [See Figure 1]