Worm Breeder's Gazette 8(3): 53
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
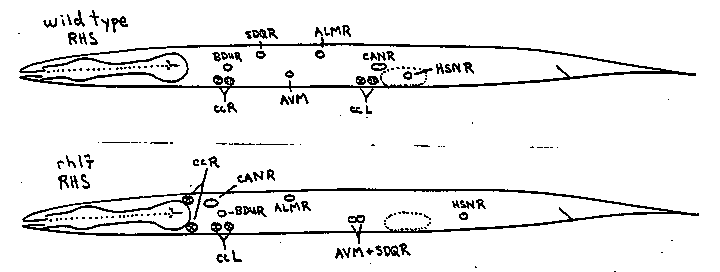
Mutations in several genes have been identified that affect the migrations of particular classes of cells. For example, vab-8 (LGV) mutations disrupt the embryonic migration of the CAN cells from their origin in the head to their final position over the gonad primordium ( Sulston and Hodgkin, Newsletter vol. 5. No. 1, p.19; Manser and Wood, ibid vol. 8, No. 2, p.9). Mutations in other genes affect the migrations of the HSN neurons, the Q neuroblasts, and the sex mesoblasts. Many of these mutants are pleiotropic but none suggest a common component to all cell migrations. A recessive mutation, mig(rh17), on linkage group X disrupts the migrations of many classes of cells. In particular, embryonic neurons ALM, CAN, and HSN are all found displaced partway toward their positions of birth. The postembryonic migrations of neurons (AVM, AQR, PQR, SDQR) derived from the Q neuroblasts are similarly affected. The embryonic coelomocytes are displaced anteriorly and the postembryonic movements of the sex mesoblasts are sometimes abnormal. A small fraction of the animals have pale, thin tails. A similar, but more severe, phenotype occurs in vab-8(e1017) mutants where it has been attributed to defective CAN migration (Sulston and Hodgkin loc cit). The rh17 mutants are also defective in egg-laying presumably because the HSN is displaced. The embryonic migrations of the gonad, the M mesoblast and its homologue (mu int R) are apparently normal. As the migrations affected by rh17 are reduced in extent but not abolished, conceivably the null phenotype is more severe. Typical cell positions for some of the affected cells in wild type and mig(rh17) mutants are shown below: [See Figure 1]