Worm Breeder's Gazette 8(1): 28
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
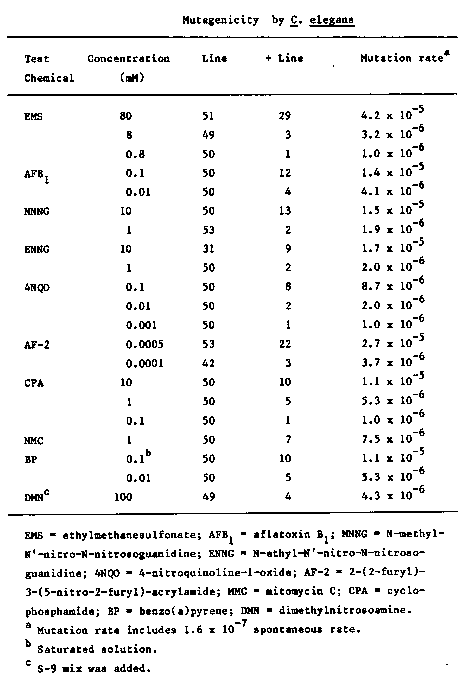
Chemical carcinogenesis is considered to proceed in the two definable steps: initiation and promotion. Detecting the corresponding initiators and promoters is thus desirable and important in carcinogenicity testing. We reported previously (Miwa et al. 1982. J. Cancer Res. Clin. Oncol., 104:81-87) that promoters such as tumor promoting phorbol esters specifically disturb development, reproduction, and behavior of C. elegans and suggested (Miwa et al. 1980. Igaku no Ayumi, :910-912) the use of this animal to screen and detect tumor promoters. Here we describe a method to test the mutagenicity of chemicals and evaluate mutagens, which are often synonymous to initiators. The method is simple, rapid, inexpensive, and reproducible and demands essentially no technical skills. It consists of treating with a given chemical a population of the animals (F0 parents) that cannot move, allowing the F0 and F1 animals to self, and scoring those animals of the F2 that move toward an attractant and away from a repellant to reach successfully a scoring position. Our current standard procedure consists of treating 200,000 L3 animals (F0 parents) of an unc tester with a given chemical for 4 hours and allowing them to self and lay eggs (F1 offspring) at 20 C. The F1's are synchronized and divided into 50 separate lines of 10,000 F1's for one concentration of a chemical . Thus, an equivalent of one million genomes that are exposed to a chemical is screened for each concentration . Each line is allowed to grow until more than 50 F2 's from each F1 mature to adult. Thus, about 10 'mobile' revertants from an F1 are expected to mature if the F1 harbors even a recessive mutation to revert unc. In order to separate revertants from the rest, we use an agar plate with pre-grown bacteria as an attractant on one side and a paper strip soaked with 1 ml of 100 mM H2S04 as a repellant on the other. The animals of each line are placed 2 cm from the bacteria and right next to the paper strip. Each line is scored for the presence (+) or absence (-) of revertants that reach the scoring position (bacterial lawn) within two hours after placement. After each line is scored, the mutation (reversion) rate per genome is calculated according to the Poisson distribution. The table shown on the following page is based on the data obtained with one of unc-13(e1091) testers and shows dose dependence of the mutation rate. Also to be noted is that our test system detects the mutagenicity of AFB1, CPA, and BP without metabolically activating treatment. These compounds require the metabolically activating treatment in the bacterial test systems such as the Salmonella test. We can carry out the whole test in 10 days, and two persons could test 15 chemicals, each at 5 different concentrations, a month. [See Figure 1]