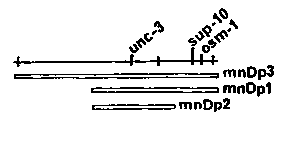Worm Breeder's Gazette 7(2): 9
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
The free duplication mnDp3(X;f) carries unc-3+ and osm-1+. About 1/1000 of the Unc-3 self-progeny of unc-3 mnDp3 hermaphrodites segregate non-Unc-3 self progeny. Six such Unc-3 animals were stained by fluorescein isothiocyanate (FITC) according to the procedure devised by Ed Hedgecock; FITC stains particular neurons of amphids and phasmids of wild-type but not osm-1 animals (Hedgecock et al., last Newsletter). Five animals were completely negative and the sixth showed staining of neurons in the left amphid only. The non- Unc-3 self progeny of all six animals stained normally. The FITC staining results thus confirm that all six Unc-3 animals were mosaic, i.e. they did not carry mnDp3 in certain somatic cells but retained it in their germ lines. If one assumes that osm-1 is cell autonomous with respect to FITC staining, the above results are easily interpreted on the basis of the known lineages (Sulston et al., last Newsletter): in five cases, duplication loss occurred in the AB founder cell and in the sixth case, loss occurred in ABp. A model in which amphidial staining depends on the genotype of the associated sheath cell or socket cell (both of which also belong to the AB line) does not fit the pattern of staining found in the sixth animal unless more than one event of duplication loss occurred in the development of that animal. In any case, the results strongly suggest that the Unc-3 phenotype is specific to AB cells. The somatic losses that gave rise to Unc-3 animals have all occurred early in the AB lineage. The reason for this could be that duplication loss is much more likely in the early divisions or that the full Unc-3 phenotype is expressed only when a distantly-related set of AB cells is mutant. To further test the idea that unc-3 expression is specific to AB, I made use of unc-93 III and sup-10 X (Greenwald & Horvitz, 1980); sup- 10 is a recessive suppressor of unc-93, which by itself confers an uncoordinated phenotype that is very different from that of unc-3 and that has been attributed to abnormal muscle structure (Greenwald & Horvitz, 1980). Because mnDp3 carries the dominant allele sup-10+, animals of genotype unc-93; sup-10; rally segregate two types of self progeny: Unc-93, with the same genotype as the parent, and Unc-3 non- Unc-93, of genotype unc-93; sup on the assumption that sup-10 is specific to muscle cells, a mosaic animal generated from the zygote unc-93; sup-10; mnDp3 is retained in AB cells but lost from body muscle cells should be non-Unc-3 non-Unc-93, i.e. wild type. Given the body muscle cell lineages, there is only one way a single somatic duplication loss will lead to a large majority of muscle cells lacking mnDp3: loss by P1, which would also lead to absence of the duplication in the germ line. Thus all self progeny of unc-93; sup-10; wild type by virtue of being somatic mosaics should give only Unc-3 non-Unc-93 offspring. Wild-type recombinants, on the other hand, should give some wildtype self progeny. I have found four mosaic animals satisfying these criteria, and the calculated frequency of loss in P1 (~ 1/2000) agrees with the frequency of loss in the sister cell, the AB founder cell, observed in the earlier experiments; thus it seems likely that the Unc-3 mosaics were also produced by single events, as already supposed. Unc-93 animals are egg-laying deficient, presumably because of defective vulval and uterine muscles. If loss of mnDp3 occurred in an unc-93; sup-10; fter formation of P1 but prior to formation of the cell that is precursor to all vulval and uterine muscles (a sequence of 9 cell divisions), the body muscles responsible for movement would be largely Unc-93, but the vulval and uterine muscles would be non-Unc-93, which could lead to egg-laying proficiency; such animals would be non-Unc-3 and retain mnDp3 in their germ lines. I have found 15 animals meeting these predictions, at a relatively high frequency (~ 1/350), which may be a consequence of the many divisions during which duplication loss may occur. As a control for the experiments involving sup-10, I have looked for both wild-type mosaics and Unc-93-moving egg-laying-proficient mosaics, as defined above, among the progeny of unc-93; )/+; sup-10 hermaphrodites. Since mnDp1 is translocated to LGV and is not free, I expected to find no mosaics. None were found. This indicates that the exceptional animals identified in the mnDp3 experiments were not simply the result of occasional incomplete expression of the single sup-10+ allele. I conclude that unc-3 expression is specific to the AB lineage and sup- 10 expression is specific to muscle cells. The free duplication mnDp26 carries the right end of X, including unc-3+, as well as various genes from LGV, including unc-60+, which affects muscle structure and confers a paralyzed phenotype (Waterston et al., 1980). An Unc-3 non-Unc-60 segregant from unc-60; osm-1; d that was completely Osm-1, by FITC staining, and gave some non-Unc-3 non-Osm-1 self progeny; this mosaic animal thus indicates that the absence of unc-60+ function in AB cells does not give the Unc-60 phenotype. The frequency of duplication loss seems to be a function of duplication size. The small duplication mnDp2 is lost ten times as often as mnDp3. And mnDp26, which seems to be large, is lost one- tenth, or less, as often as mnDp3. In screening for the relatively rare mosaic animals, I have also recovered new mutants or genetic recombinants. All cases that I have analyzed proved to involve mutant or recombinant duplications. Altered forms of mnDp1, n recovered. In a few cases, the duplication has been shown to have a deficiency. I am working to extend these studies to other genes and other duplications. I also plan to test mutations or treatments that might enhance the frequency of duplication loss; all of the work so far has involved spontaneous events. Finally, I think these results enhance the attractiveness of Bob Waterston's idea of putting sup-7 (or sup-5) on a free duplication, which could then be used to give mosaic expression of any gene that is suppressible by one dose of the suppressor.