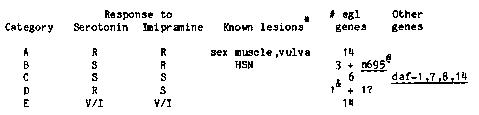Worm Breeder's Gazette 7(2): 47
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
We have characterized the egg-laying behavior of mutants defining each of 39 new egl genes (egl, egg-laying defective see Newsletter Vol. 7 No.1) as well as mutants in some previously identified genes that also affect egg-laying. Single hermaphrodites were placed on petri dishes and followed to determine: (i) whether the animal released most, some or none of its progeny, and (ii) the earliest stage of progeny ( early egg, late egg or larvae) observed on the plate. General categories were evident, ranging from mutants that lay no eggs and form bags of worms to mutants that lay only later stage (e.g. comma or pretzel) eggs or larvae but eventually release most or all of their progeny. We have also tested these mutants for their responses to the pharmacological agents serotonin, imipramine, levamisole and phentolamine, all of which stimulate the release of eggs when applied exogenously to wild-type hermaphrodites. The mutants can be grouped according to their responses to serotonin and imipramine. [See Figure 1] The only known essential components of the egg-laying system, as determined from laser ablation studies by Bob Horvitz and John Sulston, are the vulva, the sex muscles and the hermaphrodite-specific neurons (HSN's). The other neurons that provide synaptic input are not required for egg-laying. Based on the drug responses of animals with defined lesions in the egg-laying system (see Table), predictions about the nature of the defect in the egg-laying system can be made for egl mutants in categories A and B. Mutants in category A may have lost sex muscle or vulva function. Preliminary characterization by Paul Sternberg of some of these mutants indicates that at least two have defects in the divisions or migrations of precursors to the sex muscles. Almost all mutants with severe egg-laying defects (i.e. release only some or no progeny and become bags of worms) fall into category A. (eg1-2, in category D, is the single exception.) Mutants in category B are candidates for loss of HSN function. Anatomical characterization by Hilary Ellis of two of these mutants, egl-1(n487) and her-1(n695) (see next summary), has shown that both lack HSN cell bodies. Mutants in this category lay late stage eggs or larvae and release some or most of their progeny. The drug responses of mutants in categories C, D, and E do not correspond to those of animals with defects in egg-laying produced by elimination of any of the known components of the egg-laying system. Therefore, the phenotypes of mutants in these categories may result from: (i) loss of function of a yet undefined component of the egg- laying system (ii) altered function of a known or unknown component ( iii) reduction of function of a known (or unknown) component (although it seems unlikely that mutants in category D represent partial loss of function of a known component). Included in category C are four genes with temperature-sensitive dauer constitutive alleles. At 25 C or 20 C , those animals that do not form dauers form egg-laying defective hermaphrodites. These mutants may define components of the egg-laying system involved in processing environmental signals that function to modulate both egg-laying and dauer formation.