Worm Breeder's Gazette 7(2): 19
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
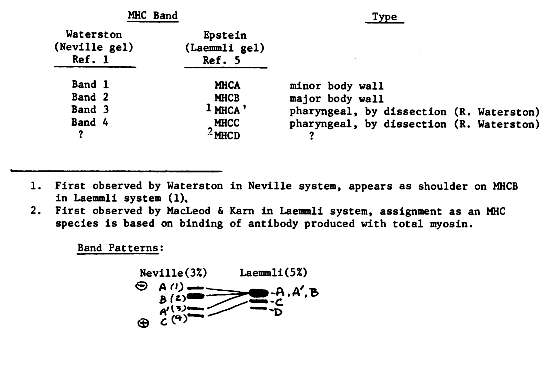
Sup-3 does not control MHCA accumulation directly. Bob Waterston's lab has reported (1) that suppression of two mutations of the body- wall myosin heavy chain major isoform (MHCB), i.e., unc-54(e190) and unc-54(e675), by sup-3(e1407) is accompanied by an increase in the MHC minor isoform (MHCA). The MHCA increase has been proposed to be a compensatory mechanism that results in an increase of the total MHC levels in sup-3 (1). The MHCA increase was mistakenly hypothesized to result from the loss of regulation of the MHCA gene which would lead to constitutive MHCA synthesis (A. Otsuka, unpublished and ref. 2). The deregulation hypothesis has been questioned by Waterston on the basis of genetic evidence (3). Due to technical difficulties, it was not previously possible to directly measure the level of MHCA in strains containing the sup-3( e1407) mutation alone. By modifying the gels used by Waterston, we have found that the MHCA levels are unaffected in CB1407 sup-3(e1407) when compared to N2. Thus, sup-3 does not directly control the MHCA level, but facilitates the accumulation of MHCA in strains with sup-3 suppressible muscle defects. It seems likely that the MHCA increase is due to an increase in the efficiency of its assembly into thick filaments. Thus sup-3 could affect some unknown thick filament assembly or MHCA modification function. MHCA levels are elevated in sup-3 Suppression of unc-54 mutants by sup-3 could result from replacement of defective MHCB by functional MHCA. However, suppression of unc-15 ( paramyosin) mutants cannot be so simply explained because the MHCB is wild type in these mutants. When we examined Sup-3(e1407) 3) animals, we found that MHCA levels were elevated. The MHCA increase in suppressed unc-15 mutants argues for a specific role for MHCA in suppression by sup-3. The possibility exists that sup-3 suppression of unc-54 and unc-15 occur by the same mechanism, a mechanism which permits more efficient assembly of MHCA into thick filaments. We have also isolated an antisuppressor of sup-3, which we call asp00(d41). d41 is a dominant antisuppressor, has no obvious phenotype alone and is not linked to either sup-3 or unc-15. Gels of total protein from asp00( d41) Sup-3(e1407) 3) animals show that MHCA levels remain elevated. Thus, d41 acts at a level that is distinct from MHCA accumulation. Gel separation of four MHC's. The myosin heavy chain isoforms were separated on a 3% acrylamide - 0.08% bisacrylamide gel (modified from refs. 1 & 4). The gel was 20 cm long (although better separation might be achieved with a 40 cm gel) and was run at 30 ma until the dye front reached the bottom (about 4 hrs). Hypochlorite synchronized L3 worms were boiled in Laemmli gel loading buffer (5) with the exception that 0.0267M H2S04-0.0541M Tris(pH6.1) replaced the glycine buffer component. In order to avoid overloading and smearing of MHCB, a limited amount of protein was used and the bands were visualized with silver stain (6). This trick permits the complete separation of four MHC species. Uncertainty as to the number of MHC species. There has been some confusion about the number of MHC species. After discussing this problem with Bob Waterston, Dave Miller, Henry Epstein, Phil Anderson, John Karn and Sandy MacLeod, the following information emerged: [See Figure 1] Thus it is still not clear whether there are more than four MHC isoforms. However, the relative amounts of MHCA & B can now be determined by a simple gel assay.