Worm Breeder's Gazette 6(1): 41
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
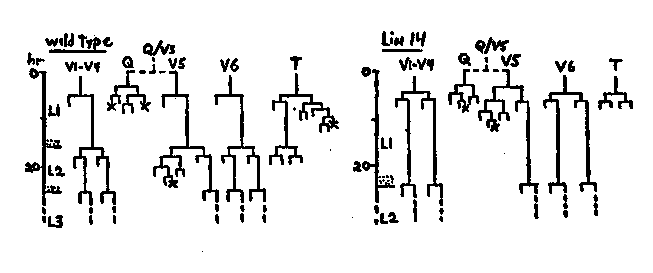
The cell lineage mutant n355 has a phenotype essentially identical to that of lin-4(e912) II, which has been described previously ( Chalfie, Horvitz and Sulston, Cold Spring Harbor Meeting Abstracts, 1979; also, Cell, in press). Aspects of this phenotype suggest that n355 and e912 repeatedly express elements of a larval (possibly the L1) developmental program: L1 hypodermal cell division patterns are reiterated during later stages; sexually mature adults have larval cuticles (based on the absence of adult-specific lateral alae and SDS gel analyses by Meredith Kusch and Bob Edgar of cuticular protein patterns); sexually mature adults undergo supernumerary molts. Unlike e912, the n355 mutation (isolated after gamma-ray mutagenesis) is dominant and sex-linked. The semidominant sex-linked mutation n536, isolated following EMS mutagenesis, results in similar but less severe defects: n536/n536 animals undergo supernumerary molts and lack adult lateral alae; n536/+ animals have normal lateral alae but have other characteristics like those of n355 and e912. All three mutants (e912, n355, n536) are long, 'floppy' and vulvaless. Revertants of e912, n355 and n536 have been isolated after gamma-ray mutagenesis, and one revertant of e912 has arisen spontaneously. Revertants have been identified by screening for egg-laying, presence of a vulva, more normal morphology and/or presence of adult lateral alae (alae can be visualized using incident illumination at 50X in the dissecting microscope). All revertants obtained thus far (two of e912, six of n355, four of n536) are themselves recessive cell lineage mutants, are allelic, and define the gene lin-14 X. n355 and n536 are very closely linked to the suppressors carried by these revertants (<0. 03% recombination) and may be within lin-14. One recessive lin-14 allele (n530) has been isolated after EMS mutagenesis of N2 and is phenotypically similar to these revertants. The defects of these recessive lin-14 alleles differ in severity. n355 n535 and n536 n540 (revertants of n355 and n536, respectively) are the most extreme. By several criteria, these mutants seem to skip aspects of the normal L1 stage. First, the cuticle of a newly hatched larva is like that of wild-type L2-L4 larvae (i.e., it lacks L1- specific lateral alae). Second, the L1 lateral hypodermal cell lineage patterns are skipped, and L2 divisions occur instead (see diagram.). V1-V6 and T all produce lineage patterns normally expressed by certain of their descendants during the L2 stage. Like the normal postdeirid sublineage generated by V5.pa in wild-type L2 animals, the mutant postdeirid-like sublineage of V5.a (as well as that of Q) produces a dopaminergic neuron. That both Q/V5 and V5 in the mutant acquire aspects of the fate associated with V5.p in wild type suggests that these three cells may be essentially equivalent in developmental potential, with their specific fates determined by developmental stage. Ventral cord development is normal, being complete at about the L1 molt. However, vulval divisions begin almost a full stage early, and vulva morphogenesis is complete at the time of the L3 molt. L4 animals are precocious adults in that they possess one or two (usually nonfunctional) vulval protrusions as well as lateral alae like those normally found only in the adult. Gonad development seems to keep normal pace with the molts, and sexual maturation follows the fourth (and final) molt. Mature adults have lateral alae. Recessive lin-14 mutations are almost completely epistatic to e912, which probably accounts for their isolation as suppressors of e912, n355 and n536. However, e912 partially suppresses the lin-14 defect of some alleles. For example, whereas lin-14(n179) hermaphrodites have adult alae during the L4, e912; n179 L4 animals do not. The accelerated development caused by recessive lin-14 mutations, which result in .skipping aspects of the L1 stage, seems to be opposite to the 'neotenic' changes produced by n355, n536 and e912, in which later developmental stages retain characters normally associated only with an earlier stage. Such alterations in the relative time of appearance and rate of development of different characteristics is known as heterochrony, which has been proposed to be a major evolutionary mechanism [See Figure 1]