Worm Breeder's Gazette 16(2): 46
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
Wellcome Centre for Molecular Parasitology, The Anderson College, The University of Glasgow, Glasgow, Scotland. G11 6NU
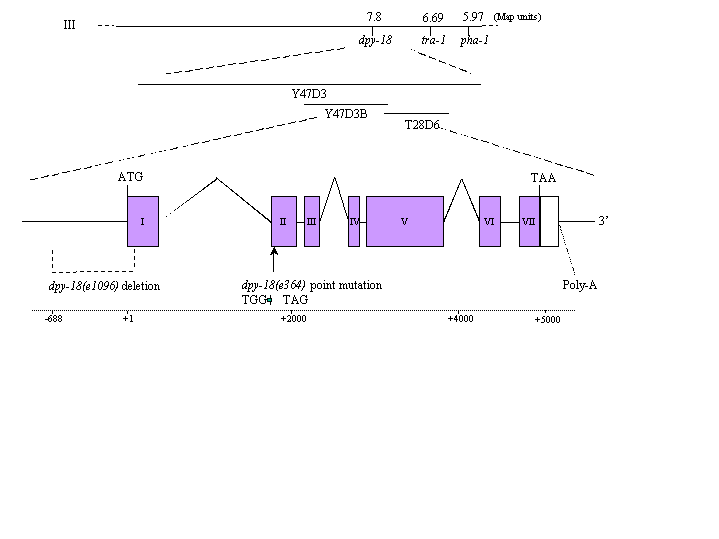
Formation of the collagen-rich cuticle is a major phylum-specific characteristic of nematodes and represents a potential target for anti-nematode drug development. Collagen molecules consist of three individual polypeptides wound around each other in a triple helical conformation. Proline residues are found at high levels in collagens (up to 24%), a large proportion of these are modified to 4-hydroxyproline which stabilizes the collagen triple helix. Collagens lacking 4-hydroxyproline have lowered melting points and are unstable at physiological temperatures. The enzyme prolyl 4-hydroxylase (P4-H) co-translationally hydroxylates proline residues in growing collagen polypeptides. P4-H occurs in C. elegans as an alpha/beta dimer (Veijola et al 1996 Biochem. J. 317: 721-729). The enzymatic activity of this complex is contained within the alpha subunit (PHY) with the beta subunit being identical to the multifunctional enzyme protein disulphide isomerase (PDI). The complete genome sequence of C. elegans has allowed identification of all sequences predicted to encode PHY. Genetic disruption of one gene from this class, phy-1 (T28D6.1), via double stranded RNA mediated interference results in a medium dumpy (dpy) phenotype. Comparison of the physical and genetic maps revealed the previously identified but uncloned dpy-18 locus to be a candidate for phy-1. Rescue of dpy-18 (e364) and dpy-18(e1096) mutant alleles was achieved by microinjection of these strains with a wild-type phy-1 clone containing 2 kb of promoter, genomic coding region and 3' untranslated region. Cloning and sequencing of phy-1 from cDNA and genomic DNA from the dpy-18 (e364) strain revealed a nonsense mutation in the second exon of phy-1, where a TGG (tryptophan) codon is mutated to a TAG amber stop codon (Figure 1). A similar approach was taken for a second dpy-18 allele, dpy-18 (e1096) which contains a 776 bp deletion of phy-1 extending from position -688 relative to the translational start and removes part of the first exon (Figure 1). This analysis demonstrates, in vivo, the critical role for prolyl 4-hydroxylase in collagen formation and maintenance of worm body shape. To date, this work represents the first described mutation affecting this class of enzymes in any organism.
Figure 1
