Worm Breeder's Gazette 16(2): 44
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
Fox Chase Cancer Center 7701 Burholme Ave. Philadelphia, PA 19111
The down-regulation of expression of the heterochronic gene lin-28 is necessary for the normal timing of larval development. This down-regulation appears to act at a post-transcriptional level because the levels of protein diminish at times when mRNA levels remain constant.
In our efforts to characterize the post-transcriptional control of lin-28 expression we observed lin-28 mRNA associated with the polysomal fraction of sucrose gradients using RNase protection assays (RPAĺs). The same finding has been made for lin-14 by Olsen and Ambros (WBG 13(5): 57). The presence of mRNAĺs in the polysomal region suggests that they are being translated by ribosomes. We now report further evidence that the mRNAĺs are being actively translated by polysomes. We first attempted to observe this by allowing ribosomes to complete translation of mRNAĺs in worm extracts and observe a corresponding shift in the distribution of mRNAĺs out of the polysomal region (this is termed translational "run-off"). We were not able to observe any run-off in worm extracts alone. We did not observe any effects in extracts incubated with the chemical puromycin, which releases mRNAĺs from translating ribosomes. However, we were able to observe run-off when we tried a new approach, by using purified polysomes in combination with a heterologous extract.
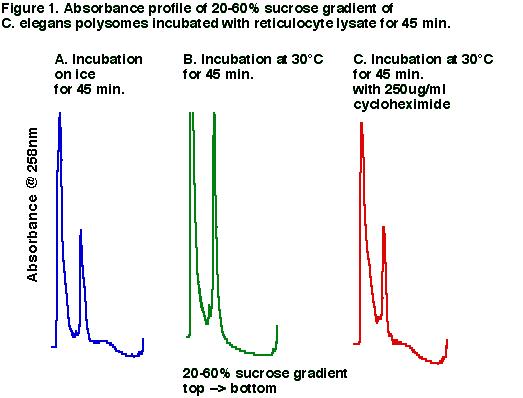
We have purified polysomes from staged C. elegans extracts, which appear to be active in translation when combined with reticulocyte lysate and amino acids (Promega). Worm extracts were prepared by grinding in a mortar and resuspending in Tris buffer containing detergents (Davies and Abe, 1995). Nuclear components were removed by centrifugation. The polysomal fraction was purified by pelleting this supernatant through a 50-60% sucrose cushion (Davies and Abe, 1995). We incubated the pelleted material with a nuclease-treated reticulocyte lysate and amino acids for 45 minutes on ice and at 30░C with and without cycloheximide. When we examined the material after incubation on 20-60% sucrose gradients we observed a shift in the absorbance profile for the polysomes incubated at 30░C without cycloheximide, but no shift for those incubated on ice or with cycloheximide (Fig. 1). Because no shift was observed in the presence of cycloheximide, a translation elongation inhibitor, we believe the shift is due to translation run-off and not to degradation of mRNA. We have repeated the experiment at 25░C and observed the same results. For a series of incubation times at 30░C the monosomal peak increases linearly with increasing incubation time, but levels off after 1 to 2 hours. We assume that re-initiation is negligible in this system. A concentration series shows that the run-off is sensitive to cycloheximide concentrations as low as 2.6 ug/ml(9uM), and the effects of cycloheximide reach 80% of the maximum at 11ug/ml. Using this heterologous system, we examined the distribution of lin-14, lin-28, and eIF4 mRNA's after run-off. We observed a shift in the distribution of all three mRNAĺs out of the polysomes from late stage worm extracts. This suggests that the polysome-associated lin-14 and lin-28 mRNA's are actively translated in late stages. This points to something other than translation elongation as responsible for the regulation of lin-14 and lin-28 gene expression. However, we are not confident that this system faithfully recapitulates what goes on in vivo. The purified polysomes may be missing important protein and RNA components that exist with the lin-28 mRNA. In addition, the in vitro translational run-off may not be sensitive enough to detect changes in the translation rate that account for changes in lin-28 expression during development.
