Worm Breeder's Gazette 16(1): 42 (October 1, 1999)
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
Division of Biology, Kansas State University, Manhattan, KS 66506
In C. elegans Wnt signaling pathways control cell polarity during embryonic and postembryonic development. In the early C. elegans embryo Wnt signaling controls the polarity of the EMS blastomere. MOM-2 is a Wnt signal produced in the four-cell embryo by the P2 blastomere, which polarizes the EMS blastomere and thereby confers distinct fates on the EMS daughters: E, which gives rise to endoderm, and MS, which gives rise to mesoderm (Rocheleau, et al., 1997; Thorpe, et al., 1997). Other components of the Wnt pathway also function in this process, including MOM-1 (Porcupine), MOM-5, (Frizzled) and WRM-1 (ß-catenin). However, mutations affecting the transcription factor POP-1 (TCF) cause the opposite phenotype, suggesting that it represses signal transduction in the absence of Wnt signaling (Lin, et al., 1995). The level of POP-1 has been shown to be higher in the anterior MS cell than it is in the posterior E cell, suggesting that Wnt signaling functions to down-regulate POP-1 levels in E, allowing Wnt-induced gene expression (Lin, et al., 1998). Wnt signaling also controls cell polarities of the B and T (both TL and TR) cells in developing larvae. A Wnt protein encoded by lin-44 is expressed in the tail hypodermal cells and affects the polarity of the asymmetric cell divisions of the more anterior T and B cells (Herman, et al., 1995). Mutations in lin-17, which encodes a Frizzled related-protein (Sawa, et al., 1996) cause a loss of cell polarity in the B and T cells (Sternberg and Horvitz, 1988), suggesting that LIN-17 may be the receptor for LIN-44 signal. No other C. elegans homologs of Wnt pathway components are known to affect the polarities of the B and T cells.
What other Wnt pathway components might be involved in B and T cell polarity? Obvious candidates include the Wnt pathway genes involved in EMS polarity. The level of POP-1 has been shown to be higher in the anterior daughters of many anteroposterior asymmetric cell divisions that occur during postembryonic development. Furthermore, in lin-17males, some sister tail seam cells had equal levels of POP-1 (Lin et al., 1998). To determine whether POP-1 levels are asymmetric in the division of the T cell, I used a monoclonal POP-1 antibody (kindly provided by Rueyling Lin) to examine the distribution of POP-1 in the T cell daughters. I found that the levels of POP-1 were higher in the anterior T cell daughters in wild-type animals and were equal in lin-17 mutants, similar to the findings of Lin et al., 1998. However, the level of POP-1 was higher in the posterior T cell daughters in lin-44 animals, suggesting that the LIN-44 WNT signal, acting through LIN-17 FZ receptor, controls cell polarity by affecting the distribution of POP-1. These results suggest a model in which high POP-1 levels specify the hypodermal, T.a, cell fate and low POP-1 levels specify the neural, T.p, cell fate.
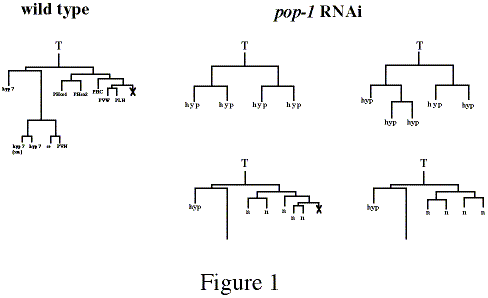
To determine if pop-1 function is required for proper T cell polarity, I wanted to examine a pop-1 mutant. However, the only existing mutation, pop-1(zu189), results in maternal-effect lethality, and has no zygotic effect on T or B cell polarities. pop-1(RNAi) also causes maternal-effect embryonic lethality, making it difficult to examine postembryonic defects. To get around this I injected double-stranded pop-1 RNA (pop-1 dsRNA) into an RNAi-resistant mutant, rde-1(ne219), isolated (and kindly provided) by Hiroaki Tabara (Tabara et al., 1999), crossed these animals to wild-type males and examined the cross progeny for defects in T cell polarity. Due to the rde-1(ne219) mutation, the maternal contribution of pop-1mRNA is not affected by the dsRNA injection; however since rde-1(ne219) is a recessive mutation, zygotically expressed pop-1 mRNA becomes susceptible in the rde-1/+ progeny of rde-1 mothers. Specifically, unc-32; rde-1 hermaphrodites were injected with pop-1 dsRNA, crossed with N2 males, and the non-Unc-32 cross progeny were scored in the late L1 stage for T cell polarity. If pop-1 functions in T cell polarity as it does in EMS polarity, and according to the model based upon the POP-1 localization results, one would predict that removal of pop-1 function would result in both T cell daughters taking on the neural T.p fate. However, I observed that in 71% (n=220) of T cell divisions both daughters expressed a hypodermal cell fate. I examined six T cell lineages through the L1 stage and observed one wild-type lineage, four lineages in which T generated only hypodermal cells (T.ap divided in one of these) and one in which T.a generated hypodermal cells and T.p generated only four neuronal cells suggesting a loss of polarity within the T.p lineage (Figure 1). Thus pop-1 zygotic RNAi results in a loss of polarity similar to that seen in lin-17 mutants. From these results I conclude that pop-1 is involved in the control of T cell polarity. Furthermore, it suggests that low POP-1 levels in T.p result in a neural cell fate, whereas loss of pop-1 function in T.p results in a hypodermal cell fate.
Literature Cited:
Herman, M. A., Vassilieva, L. L., Horvitz, H. R., Shaw,
J. E., and Herman, R. K. (1995). The C. elegans gene lin-44, which
controls the polarity of certain asymmetric cell divisions, encodes a Wnt
protein and acts cell nonautonomously. Cell, 83, 101-10.
Lin, R., Hill, R. J., and Priess, J. R. (1998). POP-1
and anterior-posterior fate decisions in C. elegans embryos. Cell,
92,
229-39.
Lin, R., Thompson, S., and Priess, J. R. (1995). pop-1encodes
an HMG box protein required for the specification of a mesoderm precursor
in early C. elegans embryos. Cell, 83, 599-609.
Rocheleau, C. E., Downs, W. D., Lin, R., Wittmann, C.,
Bei, Y., Cha, Y. H., Ali, M., Priess, J. R., and Mello, C. C. (1997). Wnt
signaling and an APC-related gene specify endoderm in early C. elegans
embryos. Cell, 90, 707-16.
Sawa, H., Lobel, L., and Horvitz, H. R. (1996). The Caenorhabditis
elegans gene lin-17, which is required for certain asymmetric
cell divisions, encodes a putative seven-transmembrane protein similar
to the Drosophila frizzled protein. Genes Dev., 10,
2189-97.
Sternberg, P. W., and Horvitz, H. R. (1988). lin-17
mutations of Caenorhabditis elegans disrupt certain asymmetric cell
divisions. Dev. Biol., 130, 67-73.
Tabara, H., Sarkissian, M., Kelly, W.G., Fleenor,J.,
Grishok, A., Timmons, L., Fire, A., and Mello, C.C. (1999). The rde-1
gene, RNA interference, and transposon silencing in C. elegans. (Cell,
In Press)
Thorpe, C. J., Schlesinger, A., Carter, J. C., and Bowerman,
B. (1997). Wnt signaling polarizes an early C. elegans blastomere to distinguish
endoderm from mesoderm. Cell, 90, 695-705.
