Worm Breeder's Gazette 16(1): 32 (October 1, 1999)
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
Department of Molecular and Cellular Biology University of Arizona, Tucson, AZ 85721
Worm fecundity is commonly assayed in many C. elegans studies. Any measurement of worm fecundity requires proper control of environmental factors that potentially affect worm fertility. In a recent article in the WBG (Twistering: A new approach to fecundity assays), Andrew Stewart mentioned that the developmental stage at which a worm is transferred significantly affected worm fecundity. Stewart reported that progeny counts recorded from worms transferred to plates as L3 worms were significantly less than progeny counts recorded from worms transferred to plates when either eggs or L2 worms. This implies that transferring older larval worms can damage the worm enough to reduce its fertility.
To examine the effect of transferring on worm fertility I measured lifetime fecundity from individual N2 worms transferred to plates as eggs, L2 and L4 worms. Worms used were from a N2 stock obtained from the CGC 3 years ago. Worms had been thawed and maintained at 20C for approximately 5 months. Worms were transferred with a platinum-wire worm-pick using a small glob of OP50. Worms were quickly transferred onto individual NGM, 35mm petri dishes spotted with OP50. A typical transfer took 5-10 seconds from when the worm was removed from the original plate to when it began crawling on the new plate. The plate was then sealed with Parafilm and placed in a 20C incubator with plate temperature and humidity monitored every 10 minutes. Fecundity counts were also done on worms that developed from an egg that was laid directly on the plate. This set of worms was never transferred and should control for any effect of transferring on worm fecundity. I assayed fecundity by either removing eggs from the plate with a platinum pick or removing worms with a pipet tip attached to a vacuum hose.
Contrary to Stewart's observations, I did not find a significant effect of transferring on worm fertility (figure 1). In conclusion, although it appears that transferring worms can reduce their fecundity, this effect can be controlled for by carefully transferring the worms.
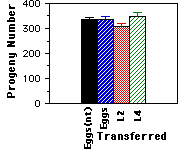
Figure 1. Average lifetime fecundity of worms transferred at different developmental stages. One group of eggs (Eggs(nt)) was laid on the assay plate while eggs from the other group were Transferred onto the plates. Progeny numbers from these four groups did not differ significantly (ANOVA, F=2.3(3,35), P=0.10. Progeny numbers and s.e.m. of worms that developed from never transferred eggs= 335 Ý8.8, developed from transferred eggs= 338 Ý9.7, developed from transferred L2 worms= 307 Ý11.7, and developed from transferred L4 worms= 349 Ý15.6. Sample size was 9-10 individuals per group. Stewart's progeny counts were: 344 progeny for worms transferred as eggs, 288 for worms transferred as L2, and 231 for worms transferred as L3's (Stewart, per. com.).