Worm Breeder's Gazette 15(5): 48 (February 1, 1999)
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
| 1 | Department of Biology, New York University, New York, NY 10003 |
| 2 | Center for C. elegans Anatomy, Einstein College of Medicine, Bronx, NY 10461 |
| 3 | david.fitch@nyu.edu |
How cells are coordinated to change their form remains a fundamental question in developmental biology. To understand the mechanism and components underlying multicellular morphogenesis, we have begun a comprehensive study of the 4-celled tail tip (hyp8-hyp11). Most recently, we have used TEM to reconstruct not only the complete cellular architecture and contacts of these cells, but also the cellular changes that occur during male tail morphogenesis.
The tail tip cells are in place by L1 and have identical cell lineages and morphologies in the different sexes. Similar morphologies are maintained until late L4, when the hyp cells fuse and retract in the male, changing the shape of the male tail from pointy to round. Fluid is excreted into the extracellular space under the L4 cuticle in the wake of the retraction. Morphogenesis does not occur in the hermaphrodite tail tip, which retains the pointy shape of the larval stages.
In "leptoderan" mutants, morphogenesis of the male tail tip fails, producing a pointy spike that protrudes ventrally and posteriorly from the adult male tail. Using this phenotype, we have identified at least 5 additional loci that appear to govern male tail tip morphogenesis. Most of these mutations (e.g., lep-1(bx37)) affect morphogenesis of hyp8-11 but not other portions of the male tail such as the fan and rays, suggesting that "general" male tail morphogenesis may be regulated independently of tail tip morphogenesis. Also, several species in Rhabditidae make good fans and ray arrays but have leptoderan tails resulting from failure in tail tip morphogenesis.
During male tail tip morphogenesis, all fusions of hyp8-hyp11 occur at sites corresponding spatially to the adherens junctions (as shown for embryonic fusions; Mohler et al., 1998, Curr. Biol. 8:1087-1090), consistent with localization of fusogenic proteins to adherens junctions. Not all the junctions correspond to fusion sites; e.g., the male-specific "hyp13" we discovered anteriorly adjacent to hyp8 (AB.p[l/r]appppa) does not fuse with hyp8-11 even though they share an adherens junction. Perhaps fusogenic components are expressed or activated in a highly localized manner. Also, fusions begin anteriorly and progress posteriorly. In lep-1 mutants, this progression is more marked because of its delay. In these mutants, hyp10, the most posterior cell, often fails to fuse and retraction fails, resulting in adults with pointy ("leptoderan") tails.
Although tail tip cell fusions and shape changes generally occur at the same developmental stage, shape changes may sometimes occur before fusions. Thus, fusions are probably not required for shape change. However, both events are delayed in lep-1 mutants, suggesting they are coordinately regulated. In other blunt-tailed ("peloderan") species such as Pellioditis typica (strain DF5025), we have found that the tail tip cells retract without fusing (see Fitch, 1997, Syst. Biol.46:145-179), suggesting that fusions and shape changes can be decoupled.
Both the fusions and the tail tip cell shape changes begin anteriorly and progress posteriorly. We think this is consistent with some triggering signal at the anterior of the tail tip. We have not yet identified the source or nature of this signal; ablations of anterior cells do not have much effect and the signal pathway mutants we have observed do not show direct effects on tail tip morphogenesis. Because some heterochronic mutants have leptoderan tail tips (M. Basson and H. R. Horvitz, pers. comm.), we speculate that the trigger and/or competence of the tail tip cells for morphogenesis requires the L/A switch governed by the heterochronic pathway, although the response to this switch would be male-specific.
Fluid is excreted into the extracellular space posterior of the tail tip cells at the beginning of retraction, but the relative volumes of the tail tip cells do not change. On the other hand, volumes of the vacuoles in the tail tip cells do appear to vary with developmental stage. Perhaps this vacuolar system (present but static in hermaphrodite tail tips) has been recruited during male tail morphogenesis to move fluid from an anterior source through the tail tip hyp cells by transcytosis.
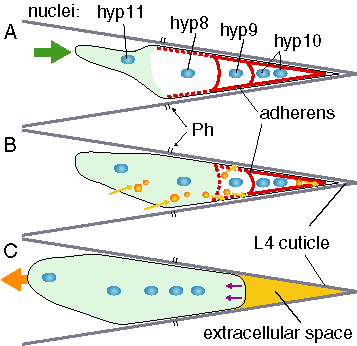
The following figure schematizes our working model for the events that occur during male tail tip morphogenesis (ventral view). First, morphogenesis is triggered, probably by a signal originating anterior to hyp8-11 (large arrowhead in A). Fusions (dashed thick lines) initiate at the adherens junctions, begin anteriorly, and progress posteriorly (A, B). Fluid is transferred through tail tip cells by vacuoles (small spheres, B) to extracellular spaces, eventually accumulating in the extracellular space of the tail spike (C). The cells (or syncytium) change shape and retract (small arrows in the posterior of C), the syncytium moves anteriorward (large arrow, C) and the nuclei are transloacted anteriorly past the phasmid openings (Ph). These events are coordinated (e.g., through a genetic pathway that may include lep-1) during the mid-L4 stage just after male-specific cell proliferations have been completed.

For more illustrations, see – http://www.nyu.edu/projects/fitch/