Worm Breeder's Gazette 15(5): 32 (February 1, 1999)
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
| 1 | Center For Basic Neuroscience, University of Texas Southwestern Medical Center, 5323 Harry Hines Blvd, Dallas, TX 75235-9148 |
| 2 | Department of Molecular Biology and Oncology, University of Texas Southwestern Medical Center, 5323 Harry Hines Blvd, Dallas, TX 75235-9148 |
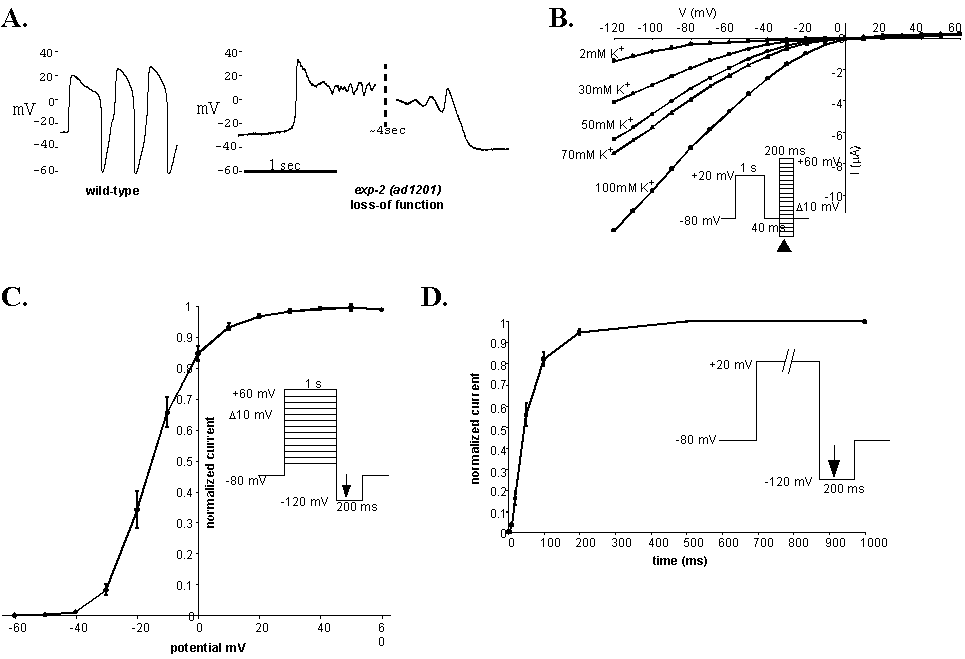
The original, semi-dominant exp-2(sa26) allele was isolated in a screen for worms with defective defecation behavior (Thomas, 1990). This gain-of-function allele also causes hyperactive head movements, defective egg laying, and brief, shallow pharyngeal contractions. Animals homozygous for this allele arrest as L1 larvae, a phenotype that can be caused by a complete inability to feed. We have isolated loss-of-function alleles which have prolonged pharyngeal muscle contraction. Current clamp recording shows the action potentials from exp-2 loss-of-function pharyngeal muscles stay depolarized for several seconds and repolarize slowly (Figure 1A) instead of the fast repolarization after ~200 ms that is seen in wild-type. We have cloned exp-2 and found that it encodes a voltage activated K+ channel. WBG 14(4): 72
Byerly and Masuda (1979) characterized a K+ current in Ascaris pharyngeal muscles that is responsible for the fast repolarization at the end of the action potential. They called the channel that carries this current the negative spike channel (NSC). Because the electrophysiological defects seen in the exp-2 loss-of-function pharynxes could be explained by loss of NSC function, we hypothesized that exp-2 encodes a subunit of the C. elegans NSC.
In order to characterize the properties of EXP-2 and see if they were indeed similar to those of the NSC, we have expressed exp-2 cRNA in Xenopus oocytes. We found that the EXP-2 channel, like the NSC and the HERG channel from the vertebrate heart, produces an inward rectifying K+ current that requires depolarization for activation (Figure 1B). Rectification occurs in all of these channels because inactivation is faster than activation. We find that the EXP-2 channel has an activation time constant of 56 ms at +20 mV (figure 1D) and an inactivation time constant that is too fast to measure. The voltage for half maximal steady state activation was -16 mV (figure 1C), similar to the NSC (-13 mV) and HERG (-21 mV). These data demonstrate that exp-2 does indeed encode a K+ channel subunit whose kinetics resemble those of the Ascaris NSC.
These properties would be well suited to repolarize the muscle quickly at the end of the action potential. The channel should be nearly fully open and inactivated at the end of a normal 150 ms action potential, and would then begin to pass current when the potential dropped enough to de-inactivate the channel. Once de-inactivation begins, K+ efflux through the channel drives the membrane potential further negative, causing even more de-inactivation and faster repolarization in a feed-forward manner.
We have not yet observed substantial outward K+ current through the EXP-2 channel, and so we are expressing cRNA from a different splice variant of the channel in the hope that this form might allow us to see such current. In addition, experiments are underway to characterize the effect of the gain-of-function mutation on channel kinetics.
Thomas, J. H. (1990). Genetic analysis of defecation in Caenorhabditis elegans. Genetics 124, 855-72.
