Worm Breeder's Gazette 15(5): 22 (February 1, 1999)
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
Department of Biology, University of Texas, Arlington, Box 19498 Arlington, TX 76019 (present address: Department of Biology, Rollins 1174, Emory University, 1510 Clifton Rd., Atlanta, GA 30322, adstewa@emory.edu)
Fecundity assays are useful for categorizing and analyzing strains of C. elegans. Unfortunately they are laborious and time consuming. Conventionally, individual worms are placed on NGM plates and picked to new plates daily and their eggs/offspring are tallied (1). Picking, however, can be damaging to worms and is a potential source of variation. Our tests have shown that N2 worms picked at the L3 stage had significantly fewer offspring than those picked as eggs (T 0.05 [14] = 4.71 p < 0.001) or even those picked at the L2 stage (T 0.05[14] = 2.55 p = 0.023). To avoid this problem, individual worms were picked as eggs onto NGM plates and the eggs/offspring were picked daily and tallied. This eliminated the physical damage of picking, but it also greatly increased the time required to score plates as experienced "pickers" could spend up to an hour per plate. To speed the process, I have devised a novel, inexpensive piece of equipment called the "Twister Apparatus" which reduces the scoring time by 60%-75%. In addition to timesavings, even novice "pickers" are capable of impressive speed while "Twistering" and there is little adjustment time required. Twistering was chosen as the name for this method (think of a tornado carrying off the worms) when I got tired of saying that I was going to go "suck worms." It must also be noted that there is another way to avoid physical damage to worms that involves mouth pipetting individual worms from plate-to-plate using a drawn out melting point capillary (2). However, this is also time-consuming.
At its most basic, the Twister apparatus is simply a worm vacuum. It is a length of flexible tubing connected to a suction source at one end and a nozzle at the other. Fifty microliter micro-pipets are ideal for Twister nozzles. They can be pulled out either by a pipette puller or by hand, although the latter takes considerable practice. The terminal end of the nozzle should be about 0.5 mm in diameter. The nozzle is then placed into a holder fashioned from two disposable pipete tips (Figure 1); yellow 200ml tips work the best (we use Fisherbrand). The tip of one is cut back so that the shaft of the nozzle will just fit inside. The wide end of the other is cut to allow it to fit inside the wide end of the other tip, held in place by friction. This assembly creates a stable holder for the fragile glass rod. This holder is then placed into the length of tubing connected to the suction source.
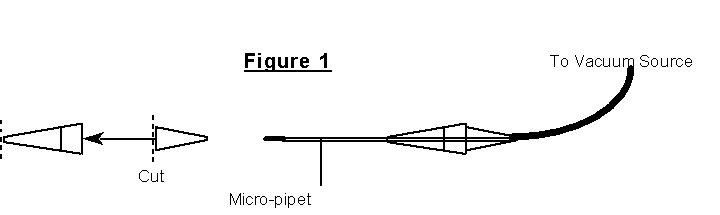
Nozzles can be cleaned by submersion in NaOH to dissolve any worms clinging to the inside and then sterilized by submersion in ethanol prior to use and as needed during Twistering. Do not flame these nozzles, as the heat will melt the thin tips.
Twistering plates has no significant effect on fecundity (T 0.05[10] = -1.36 p = 0.204) and does not damage the surface of the plate; only small spots are seen where the E. coli lawn is vacuumed up with the egg/worm and these grow closed overnight.
(1) Brooks, A and Johnson, T. 1991. Heredity 67:19-28. (2) Wood, W. B. (ed.). 1988. The Nematode Caenorhabditis elegans. Cold Spring Harbor Laboratory.