Worm Breeder's Gazette 13(4): 64 (October 1, 1994)
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
Waksman Institute and Department of Molecular Biology and Biochemistry, Rutgers University, Piscataway, NJ 08855.
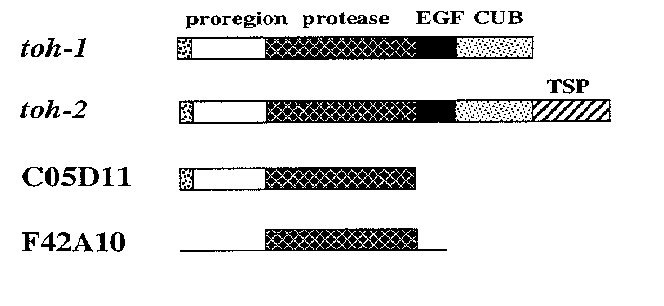
Members of the tolloid/ BMP-1 family of metalloproteases are thought to interact with extracellular growth factors to regulate a variety of developmental events. In particular, the Drosophila gene tolloid interacts with decapentaplegic (dpp), a member of the TGF-337 superfamily. Similarly, vertebrate BMP-1 co-purifies with other BMPs (bone morphogenetic proteins) that are related TGF-337-like ligands. Proteins in the tolloid/BMP- 1 family share a similar structure: they contain a predicted metalloprotease domain; EGF repeats; and CUB repeats, which are also found in blood complement proteins. These proteases are thought to interact with other proteins through the EGF and CUB repeats, and to regulate their activity through proteolysis. We have previously reported sequence analysis of the first C. elegans member of this metalloprotease family (WBG 12(4), p.30), which we have called toh-1 (tollish). Since then, the genome sequencing consortium has identified four more predicted ORFs with sequence similarity to tolloid. We have therefore isolated and sequenced cDNAs for these genes, to determine whether these new genes encode true members of the tolloid/ BMP-1 family. One of the putative homologs displayed no protease homology. The predicted structures of the other three homologs are summarized below. Two of these, on the cosmids C05D11 and F42A10 ,contain related metalloprotease domains, but none of the characteristic protein interaction motifs of the tolloid/ BMP-1 family. Only one homolog, on the cosmid R151 ,was found to contain an EGF repeat and a CUB repeat. We therefore propose the name toh-2 for this gene. toh-2 represents a novel member of this family, because it contains repeats (TSP repeats) found in thrombospondin, an extracellular matrix protein. The presence of these two tolloid homologs in the worm raises several interesting questions: (1) Do these homologs act together in a TGF-337-like signal transduction pathway, in separate pathways, or combinatorially with other, as yet unidentified, tolloid-like proteins? (2) What roles do these genes have in nematode development? (3) What is the function of the toh-2 thrombospondin repeats? Are there homologs in other organisms that also contain these repeats? (see figure)