Worm Breeder's Gazette 13(3): 54 (June 1, 1994)
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
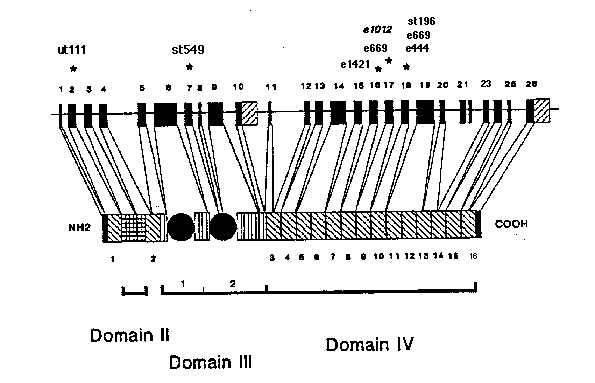
The unc-52 gene produces several large and complex proteins that function in the basement membrane underlying muscle cells. These extracellular proteins play an important role in directing myofilament assembly, and in maintaining the ordered structure of the myofilament arrays. The longest potential open reading frame of the unc-52 gene encodes a 2482 amino acid protein with four recognizable domains (Fig. 1). The first domain is unique to the UNC-52 polypeptide whereas the three remaining domains contain sequences found in the LDL-receptor (domain II), laminin (domain III) and N-CAM (domain IV) (Rogalski et al. 1993 Genes and Dev. 7:1471-1484).
Mutations in the unc-52 gene affect myofilament organization in the body wall muscle cells. Mutants homozygous for the e444 , e669 , e998 , e1012 , e1421 and st196 alleles are viable. They are able to move normally as larvae, but adult hermaphrodites are paralyzed. In contrast, the st549 and ut111 mutations result in a lethal terminal phenotype. Embryos homozygous for these alleles have very disorganized muscle cells, and fail to elongate past the "two fold" stage (Williams and Waterston 1994 JCB 124:475-490). The st549 mutation fails to complement all other unc-52 alleles whereas ut111 and the "viable" alleles complement (Gilchrist and Moerman 1992 Genetics 132:431-442).
We have identified the molecular alterations responsible for the different unc-52 mutant phenotypes (Gilchrist and Moerman, WBG 12(3):35; Fig. 1). The mutations that produce an Unc terminal phenotype are clustered in a small interval containing three adjacent, alternatively spliced exons. Four are point mutations which introduce stop codons into exon 17 ( e669 and e1012 )or exon 18 ( e444 and e998 ),one ( e1421 )is a point mutation which alters the splice donor of exon 16, and the remaining allele carries a Tc1 element in exon 18. These mutations affect some, but not all of the UNC-52 proteins. An analysis of RT-PCR generated cDNAs has revealed that as many as seven different transcripts are produced by the alternative splicing of exons 16, 17 and 18. The st549 allele carries a premature stop codon in exon 7 which should result in the complete elimination of unc-52 gene function, and the ut111 allele has a Tc l transposon inserted into the second exon of the gene.
Several intragenic revertants of the e669 , e998 , e1 O12and e1421 alleles had previously been isolated as part of a genetic characterization of the unc-52 locus (Gilchrist and Moerman 1992 Genetics 132:431 442). We have sequenced PCR amplified DNA from twelve of these revertant strains. In all but one, reversion of the mutant phenotype is apparently due to a point mutation in a splice acceptor site; usually, but not always, the splice acceptor of the affected exon (Gilchrist and Moerman WBG 12(3):35). Preliminary results from RT-PCR experiments suggest that transcripts carrying the mutated exons are never (or only rarely) made in these strains. Thus, the mutations in the splice acceptor sites of exons 16, 17 and 18 may prevent these exons from being included in any of the unc-52 transcripts. We have also sequenced PCR amplified DNA from animals carrying su250 which is a spontaneous revertant of e669 (MacKenzie et al. 1978 Cell 15:751-762). The only sequence alteration detected, aside from the e669 point mutation in exon 17, was a single base change in the adjacent upstream intron.