Worm Breeder's Gazette 13(3): 40 (June 1, 1994)
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
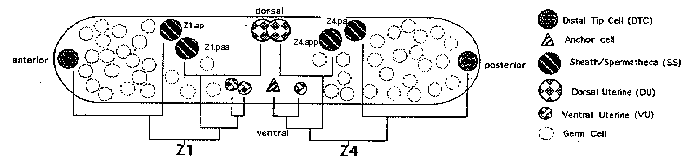
During C. elegans hermaphrodite development, germ cells complete multiple events to become sperm or oocytes. These events include mitotic proliferation, a switch from mitosis to meiosis, progression through meiotic prophase, meiotic arrest and the meiotic divisions, as well as sex determination and gametogenesis. Importantly, the germ cells share residence in the gonad with developing somatic cells which become the distal tip cells (DTCs), sheath, spermatheca, and uterus. As evidenced by DTC control of germ-line proliferation via GLP-1 ,cells of the somatic gonad can regulate germ-line development(1), (2). By analogy, additional steps in germ-line development may also fall under somatic control
We have used laser ablation to investigate the role of other somatic gonad components during hermaphrodite germ-line development with an emphasis on lineages leading to the ten somatic sheath cells that surround the germ in each arm. During L2 ,all somatic cells, except the DTCs, move to the center of the gonad forming the somatic primordium. (See Figure.) Four of these cells (Z1.ap and Z1 .paaanteriorly, Z4 .paand Z4 .appposteriorly) give rise to the entire sheath and most of the spermatheca(3). Elimination of these 'SS' (sheath/spermatheca) cells results in the three germ-line phenotypes discussed in these articles (parts I, II, III), which update our preliminary studies presented at the 1993 Worm Meeting(4).
Germ-line Proliferation Defective (Glp)
Ablation of both SS cells in one arm results in a miniature gonad arm (100% Glp, n=30). Although a distal mitotic population is maintained, proliferation is greatly reduced (257±85 germ cells per arm, 60-90 hrs. post hatch) compared to wildtype (~800). This germ-line proliferation defect is distinct from that seen in glp-1 mutants or DTC ablations where all germ cells differentiate leaving no stem cell population. The miniature arms are also defective in differentiation and are sterile (Many did not make sperm, and those that did made far fewer than normal; oogenesis was not observed). Arms have the usual U-shape and the gonad basement membrane is intact so that no contents leak. Further, there is no evidence of alterations in other lineages to replace the missing cells. Miniature arms are not seen following ablation of one SS cell, ablations of combinations of dorsal and ventral uterine cells, AC ablation, or ablation of germ cells.
Conclusions From these ablations, we conclude that SS cells or their descendants are promoting germ cell proliferation. This promotion may be via a specific signal ( GLP-1 dependent or independent), or due to a less specific trophic effect.
(2) Austin and Kimble. Cell, 51:589-99, 1987
(3) Kimble and Hirsh. Dev. Bio., 70:396-417, 1979
(4) McCarter, et. al. 1993 Worm Meeting, p.304.