Worm Breeder's Gazette 13(3): 20 (June 1, 1994)
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
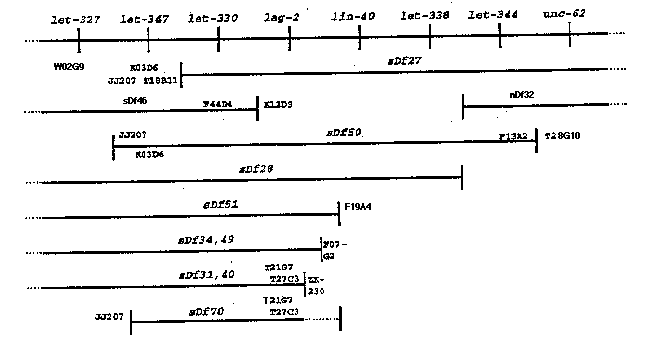
In the early stages of cloning lag-2 ,we were faced with a huge contig with no anchors to the genetic map, but with the knowledge that our answer was probably in those three megabases somewhere. We took advantage of the bounty of deficiencies, made and analyzed by David Baillie's lab, to align the contigs and the genetic map. Briefly, we made DNA from deficiency heterozygote strains (using eT1 marked with a lethal to balance deficiencies) and probed Southern blots containing several different deficiencies with probes from throughout the three contigs likely to contain lag-2 .We were able to strip and reprobe the filters (Nytran) numerous times (up to 12 and still usable).
The biggest obstacle was the presence of repetitive DNA in the cosmids used as probes. The progress of the cDNA sequencing project will provide for many more single copy probes for people using this approach. One step that we took to screen out cosmids and fragments containing highly repetitive DNA was to blot digests of cosmids and probe with labeled genomic DNA. Exposure times for these filters ("reverse Southerns") are similar to genomic DNA blots. This technique was useful for detecting highly repetitive sequences, but some low or middle repetitive sequences were found among fragments that passed this screen. Of the thirteen original cosmids we chose to test from LGV(left) contigs, nine were found to contain highly repetitive elements. There were usually only one or two dark bands per Hind III digested cosmid, allowing the identification of other fragments that could be used as probes.
For those who might find the results of our alignment of LGV useful for mapping, results with cosmids and deficiencies are shown below. Cosmids are shown outside or within deficiencies; order with respect to the genes in the deficiency interval is not known, except that T21G7 and T27C3 rescue lag-2 .Note also the results of McKim et al., WBG 12:1, p. 18. who mapped deficiencies to the left of this region using PCR. We did not map the right breakpoint of sDf70 . K03D6 was not tested with sDf70 ,and W02G9 was not tested with sDf50 .