Worm Breeder's Gazette 13(1): 86 (October 1, 1993)
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
Filariasis is a parasitic disease infecting over 300 million people worldwide resulting in Iymphoedema, elephantiasis, or blindness. The disease is caused by several species of helminthic worms (Brugia malayi, B. timori, Wuchereria bancrofti and Onchocerca volvulus) transmitted by mosquitoes and flies. A related disease, dog heartworm is caused by Dirofilaria immitis infection in the heart and lungs of dogs.
The role of ecdysone during insect metamorphosis and the superficial similarity between development of insects and nematodes through a series of larval molts to the adult, has led to the theory that molting in nematodes may also be controlled by steroid hormone(s). The presence of ecdysone has been reported in D. immitis, (Cleator et al. 1987, Mol Biochem Parasitol 25:93-105) and ecdysone has been shown to stimulate formation of microfilaria and induce the third- to fourth-stage molt in vitro (Barker et al. 1990, Invertebrate Reprod Devel 18:1-11; Barker et al. 1991, Parasitol Res 77: 65-71). However, attempts to elucidate a biosynthetic pathway for ecdysteroids in D. immitis (Mercer et al. 1989, Trop Med Parasitol 40:429-433), C. elegans (Chitwood et al. 1990, J Nematology 22 :598-607) and other nematodes have been unsuccessful. If ecdysone is being utilized in nematodes, the receptor for the hormone will be present. The Drosophila ecdysone receptor (EcR) is a member of the nuclear hormone receptor (NHR) superfamily, a group of hormone regulated transcription factors, and has been cloned and sequenced (Koelle et al. 1991, Cell 67:59-77).
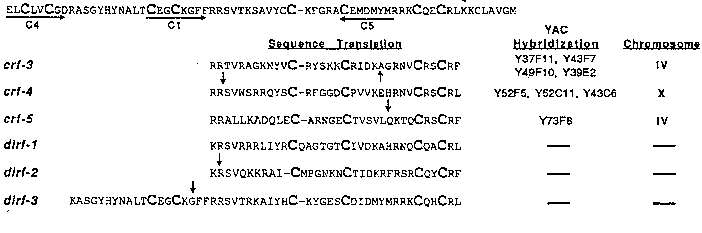
We have used the Drosophila EcR to search for D. immitis and C. elegans homologs using several approaches, the most fruitful being PCR with degenerate primers to the DNA binding domain. Four degenerate primers (C1, C2 , C4 ,and C5 )encoding the amino acids shown in the figure were used in various combinations in PCR experiments with genomic DNA from both D. immitis and C. elegans (see figure below and expanded figure in abstract by Hough et al., this issue). All PCR products were cloned into pUC19 and sequenced.
PCR with primer combination C2 /C4amplified a DNA fragment of 473 bps from D. immitis (dirf-3) that upon sequencing shows 90% similarity at the amino acid level to the Drosophila EcR. It contains a putative intron of 362 bps at the position indicated by the arrow which is within the C1 primer and is most likely why it was not amplified with the C1 /C2primer combination (see below). The Drosophila EcR does not contain an intron within the DNA binding domain. Primer combination C4 /C5amplified a single DNA fragment of 455 bps from D. immitis that upon sequencing showed it to be a smaller piece of dirf-3.
The C1 /C2primer combination amplified 3 DNA fragments from C. elegans ( crf-3 , crf-4 ,and crf-5 ,in keeping with the nomenclature for nematode NHR s established by Sluder et al. WBG 11, #3) and 2 from D. immitis (dirf-1 and dirf-2), ranging in size from 160 to 524 bps. All 5 show significant similarity to the NHR superfamily at the amino acid level, although no striking similarity to any one particular member. Four contain putative introns of sizes ranging from 45 to 258 bps at the sites indicated by the arrow.
None of the 3 C. elegans NHR s have been described previously. They have been mapped to the genome (see figure) and we have obtained cDNA s for each from a screen of a mixed stage cDNA library (Stratagene). The 3 D. immitis NHR s are the first to be described for this nematode and, to date, we have obtained a cDNA for one (dirf-2) from a screen of an adult female cDNA library (Grandea et al. 1989, Mol Biochem Parasitol 35:31-42).
Our search for a C. elegans EcR homolog continues.