Worm Breeder's Gazette 13(1): 84 (October 1, 1993)
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
The ABA-1 allergen of Ascaris suum and Ascaris lumbricoides has attracted attention for the following reasons: l) it is the most abundant protein in the pseudocoelomic fluid of the adult parasites and it is also released by the tissue-invasive larval stages 2) it is a demonstrated allergen, 3) infected humans respond heterogeneously to ABA-1 and individuals may thereby vary in their immunopathological reactions to the infection, and 4) the specificity of the antibody responses of Ascaris -infected rodents is under genetic control, the class II region of the major histocompatibility complex containing the loci involved (1,2).
Sequencing of several cDNAs encoding ABA-1 ,selected from a lambda-ZAP expression library with antibody to the protein, showed that the aba-1 gene comprises tandemly repeated units encoding the ABA-1 protein (3). Whilst some members of the array encode polypeptides of identical or near identical amino acid sequence, some are substantially different, one being only 49% identical at the amino acid level (unpublished). While work on the Ascaris protein was in progress, cDNAs encoding ABA-1 -likeproteins from filarial nematodes including Brugia malayi and Dirofilaria immitis had been cloned and sequenced (4,5.6), all being found to possess the unusual gene organization described above. In each case, the gene is transcribed and translated to form a long polyprotein which is then proteolytically cleaved to produce multiple copies of the protein monomer. This led to the coining of the term 'nematode polyprotein allergens' (NPAs) for the ABA-1 -typeproteins (2). The N-terminus of the processed molecule was established for ABA-1 by direct amino acid sequencing of parasite-derived material (7) and cleavage of the polyprotein is thought to occur at a tetrad of four arginines, similar tetrabasic sites being found in all the homologs. Further, the gene is constitutively expressed in all stages and tissues of Ascaris examined (H.J. Spence, J. Moore & M.W. Kennedy, unpublished). Molecular modeling and biophysical analysis of ABA-1 protein has led to the prediction that it falls into the four-helix bundle class of proteins which, for ABA-1 and probably all the NPAs, forms a homodimer.
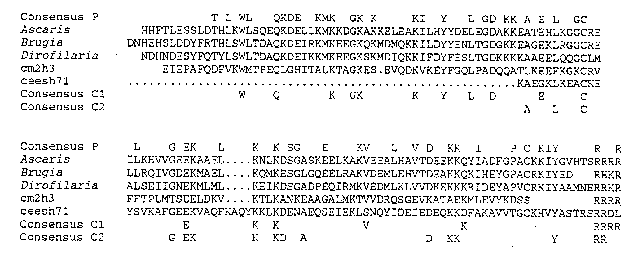
ABA-1 -typeproteins, therefore, are produced by a wide range of parasitic nematodes, but neither their biological function, nor whether they are confined to parasitic nematodes, is known. Data base searches revealed no similarity with any other type of protein, and motif searches for functional regions were similarly fruitless. On closer inspection of C. elegans databases, however, our attention was drawn to cDNA clones cm2 h3 , cm13 h12 (Cambridge& Washington) & c eesh71 (T.Kerlavage, Gaithersburg), which showed the highest level of sequence similarity with NPAs, although this was still below that which is generally thought to be significant. With knowledge of the conserved residues within NPAs of parasites, it became clear that these cDNAs did indeed represent proteins belonging to the ABA-1 family (see Figure 1). The sequences from the three C. elegans clones are different from one another, paralleling the diversity in the sequence of individual repeats encoded within the same cDNA in some parasitic nematodes; this is shown to greatest effect by Dictyocaulus vivivarus in which adjacent repeal units can be as little as 38% identical at the amino acid level (C. Britton, J.G. Gilleard & M.W. Kennedy, unpublished data). D. viviparus is considered to be phylogenically closer to C. elegans than the other parasites investigated, so perhaps represents the condition in free-living antecedents of parasites.
Two of the cDNAs ( cm2 h3 & cm13 h12 )have been mapped to chromosome V, and are contained in YAC clones 57E12 and 58H5 and cosmids F27B10 and F27A2 (Cambridge), the other cDNA hybridizes to YAC clones Y9G9 , Y6H4 & Y53G8 (YAC polytene chromosome blots kindly provided by Alan Coulson, Cambridge) and thus lies on the left arm of chromosome III. There thus appears to be two NPA-encoding loci in the C. elegans genome instead of the single locus so far apparent for the parasitic nematodes. In an attempt to elucidate the function of NPAs we intend to further characterize these cDNAs and the corresponding genetic loci with a view to generating Tc1 mutants. To date, there are no mutants known in the region defined by the YAC clones 57E12 & 58H2 which would help define a phenotype. Whatever the outcome, the NPAs present an
example of an important antigen of parasitic nematodes whose genetics, function and expression can be investigated in C. elegans. They certainly represent an unusual gene organization encoding a structurally conserved protein which may be unique to nematodes.
References
1. Kennedy, M.W. (1989) Parasitology Today, 5, 316-324.
2. McReynolds. L.A,. Kennedy, M.W. & Selkirk, M F. (1993) Parasitology Today, November issue, in press.
3. Spence, H., Moore, J., Brass, A.& Kennedy, M.W. (1993) Mol. Biochem. Parasitol. 57, 339-344.
4. Culpepper, J., Grieve, R., Friedman, L., Mika-Grieve, M., Frank, G. & Dale, B. (1992) Mol. Biochem. Parasitol. 54, 51-62.
5. Poole, C., Grandea, A., Maina, C., Jenkins, R., Selkirk, M. & McReynolds, L. (1992) Proc. Natl..Acad. Sci. USA 89, 5986-5990.
6. Tweedie, S., Paxton, W., Ingram, L., Maizels, R., McReynolds, L., & Selkirk, M.E. (1993) Exp. Parasitol. 76, 156-164.
7. Christie, J.F., Dunbar, B., Davidson, I. & Kennedy, M.W. (1990) Immunology 69, 596-602.