Worm Breeder's Gazette 13(1): 74 (October 1, 1993)
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
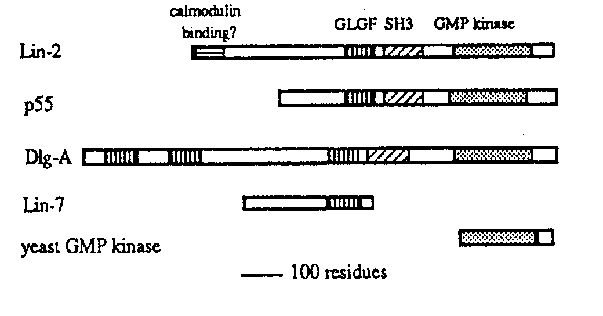
The P(5-7).p cells are induced to undergo vulval development by a signal from the anchor cell. The inductive signal is transduced by a receptor tyrosine kinase/ras pathway. Iin-2 functions in this pathway: null mutants have a 95% penetrant vulvaless (Vul) phenotype. We have found that lin-2 encodes a protein with an SH3 domain and a guanylate kinase domain, similar to the product of the Drosophila tumor suppressor discs-large (dlg). What role might a guanylate kinase play in a ras pathway?
I have cloned the lin-2 gene. The cosmid C43C10 is tightly linked to lin-2 ,complements the Vul phenotype in germline transformants, and reveals rearrangements in four alleles. n397 carries a 7.5 kb deletion that removes most of the gene, including the putative initiator codon, so it is very likely a null mutation. n397 mutant animals are viable and 95% Vul. The identification of the gene is further supported by sequence analysis of a mutant. Direct sequencing of genomic PCR products reveals a glutamine to ochre nonsense mutation in n105 ,22 residues from the end of the predicted lin-2 protein.
Sequencing of two independent cDNAs reveals an ORF with homology to dlg and p55 ,a palmitoylated, membrane-associated protein that co-purifies with a serine kinase and the actin-bundling protein dematin. The homology comprises three domains: a GLGF repeat of unknown function, an SH3 domain, and a domain homologous to yeast guanylate kinase. In some 400 residues, p55 is 51% identical to lin-2 and dlg is 34% identical to lin-2 . Iin-2 also encodes an N-terminal domain of 67 residues with 40% identity to the calmodulin binding domain of rat type II calmodulin-dependent protein kinase. Iin-7 ,which is very similar genetically to lin-2 ,also encodes a GLGF domain (Simske and Kim, this issue).
Mutations in dlg lead to non-epithelial neoplastic overgrowth of the fly imaginal discs. Thus, dlg is required to repress cell proliferation. This seems opposite to lin-2 ,which functions in induction of the vulval cell lineages. However, the genes may play similar roles in signaling pathways that simply have different consequences. That is, signaling may control when and where a cell adopts a pre-programmed fate, rather than specifying cell fate directly. dlg protein is localized to septate junctions in disc epithelia
Guanylate kinase catalyzes the transfer of a phosphate group from ATP to GMP, yielding ADP and GDP. The lin-2 and dlg proteins and p55 all have 30-35% sequence identity with guanylate kinase across the length of the yeast enzyme. In addition, all of the residues known from x-ray crystallographic studies to contact GMP in the yeast enzyme are conserved or conservatively substituted in all three proteins.
Two observations suggest that lin-2 is not the major cytosolic guanylate kinase, but rather interacts specifically with let-60 ras. First, lin-2 null mutations do not appear to affect cellular viability. Loss of the major guanylate kinase activity would lower the bulk concentrations of GDP and GTP, which are important for myriad processes in the cell. But in lin-2 mutants, the transformed Pn.p cells appear healthy and undergo an active non-vulval developmental program, including a round of cell division. Second, lin-2 acts upstream of let-60 ras and is required specifically for normal ras activation. Iet-60 ( n1700 )is a gain-of-function Muv allele that encodes a constitutively activated mutant ras protein. Iet-60 ( n1700 ); lin-2 ( e1309 , n768 or n1610 )animals are Muv, thus activated ras can override all lin-2 defects.
ras proteins are activated by GTP binding. In one model, lin-2 mutations might disrupt signaling by reducing the local concentration of GDP and hence GTP, perhaps in a special pool a protein complex, impairing the formation of active ras protein. This model predicts a ras-associated kinase converting GDP to GTP. In another model, lin-2 functions to remove GDP from the vicinity of ras protein. ras proteins have an intrinsic, inactivating GTPase, which hydrolyzes bound GTP to GDP. The GDP-bound protein is not active in signaling. ras protein then exchanges GDP for GTP to "reload." The affinities of ras protein for GDP and GTP are similar, so if the concentration of GDP is too high, ras is inhibited. In this model, lin-2 prevents a local buildup of GDP by converting it to GMP, and thus prevents inhibition of ras. A third possibility is that lin-2 is required indirectly, for cell polarity or adhesion, to facilitate signaling. We are devising tests of these models.