Worm Breeder's Gazette 13(1): 42 (October 1, 1993)
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
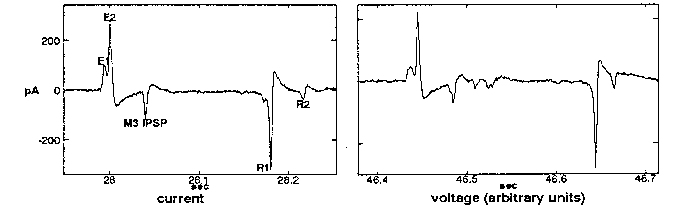
When a worm's head is sucked into a pipet and the inside and outside of the pipet connected to a high-impedance voltage amplifier, voltage transients are measured between the inside and outside of the pipet every time the worm pumps (Raizen and Avery, WBG 12(5): 62). Although the time-course of these transients is quite reproducible from pump to pump and from worm to worm, their size varies from less than a few hundred µV to several mV, even in the course of a few minutes' recording from a single worm. We suspected that this variability was an artifact of our recording arrangement; that in fact each transient in the electropharyngeogram (EPG) carried roughly the same current in every pump, and that the variable voltage we measured resulted when this current flowed across the variable resistance of the seal between the pipet mouth and the cuticle of the worm's head. To test this hypothesis, I connected the pipet to a low-impedance current-measuring amplifier (an Axopatch 1D patchclamp amplifier operated in voltage-clamp mode). As hoped, the transients were now constant in size, not only from pump to pump, but even among worms of the same age and size. A typical record of a single pump is shown below (left), accompanied by a typical voltage trace for comparison (right). Positive currents represent current flowing out of the worm's mouth. All the usual features can be recognized.
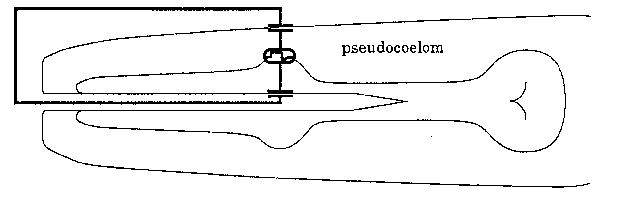
What is the origin of this current? When pharyngeal muscle is excited, the voltage changes across the basal membrane, which separates the muscle cell cytoplasm from the pseudocoelom. The total voltage difference between the muscle cytoplasm and pseudocoelom must be the same alone any path that connects them, including the long path that goes across the apical membrane of the muscle cell into the pharyngeal lumen, out the mouth, through the surrounding liquid, across the cuticle and hypodermis, and back into the pseudocoelom (see diagram above). Most of the voltage drop along this path should appear across the membranes, since cytoplasm and pseudocoelom have low resistance. Biological membranes are capacitors: any voltage difference between the two faces of a membrane is accompanied by a proportional difference in charge. Thus, when pharyngeal muscle is excited, current must flow to charge the apical membranes and hypodermis. If we assume that all the charging current flows out of the mouth, and that most of the voltage drop appears across the apical muscle membrane instead of the hypodermis (equivalent to assuming the capacitance of the apical membrane to be much smaller, reasonable since capacitance is proportional to area), we can estimate roughly the voltage change associated with each transient.
This analysis was done using the R1 spike, since it is large and well-isolated, and since we understand it best. The R1 spike results from the repolarization of the corpus muscle that takes place at the end of an action potential. The total charge (measured as the area of the trough) transferred during the R1 spike of a healthy adult N2 hermaphrodite was close to 1 µF/cm(2). From the pictures in Albertson and Thomson's reconstruction of the pharynx (Phil Trans Roy Soc 275B: 299-325), the area of the apical membrane of the corpus can be very roughly estimated at 2000 µm(2). The capacitance of biological membranes is usually close to 1 µF/cm(2). These numbers give an estimated voltage change of 50 mV. Byerly and Masuda (J Physiol 288: 263-284) measured a voltage change of 100µmV when Ascaris pharyngeal muscle repolarizes. Considering the uncertainties in the calculation, this agreement is remarkable, and suggests that much of the current that flows out of the mouth arises as described.
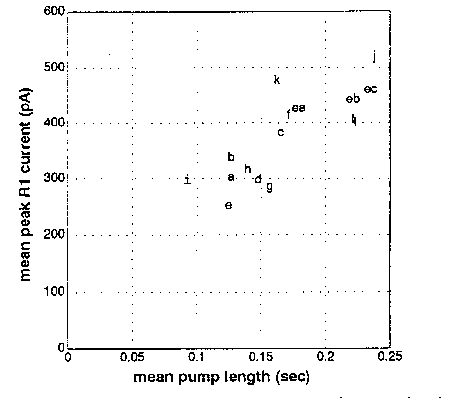
In the course of this analysis, I noticed a correlation between the length of the pump (i.e., the interval from E2 to R1 spike) and the depth of the R1 spike. The figure below shows pump length and R1 depth for 12 N2 adults (a -1) and 3 egl-30 adults (ea - ec; egl-30 worms tend to have longer pumps). The correlation could also be seen among the pumps recorded from a single worm. This correlation may be explained by the properties of the Ascaris negative spike channel described by Byerly and Masuda (ibid) and believed to mediate the repolarization at the end of the action potential. This channel is inactivated at resting potential, and the inactivation is removed by depolarization with a time constant in the range of hundreds of msec. (The precise time constant depends on membrane potential.) Thus, the longer the muscle cell remains excited, the more negative spike channels will be available to open when the action potential ends, and the larger the peak current will be. The increased current is expected to speed up the repolarization of the membrane, but should have little effect on the total voltage change. In fact, the deeper troughs tended to be narrower so that the total charge transferred (which, as described above, should be proportional to the change in membrane potential) didn't vary as much as the peak current.
In summary, the currents that flow out of the mouth due to pharyngeal activity are of reproducible amplitude. The total charge that flows into the mouth during corpus repolarization is of about the order of magnitude predicted from simple electrical considerations. Finally, the peak current during corpus repolarization is correlated with the length of the action potential, a result that can be explained by the hypothesis that the negative spike channel of Byerly and Masuda passes this current.