Worm Breeder's Gazette 12(5): 57 (February 1, 1993)
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
Biochemical and pharmacological studies have revealed that ion transport across the C. elegans sperm plasma membrane probably accompanies formation of spermatozoa (Nelson and Ward, Cell 19: 457-464, 1980; Ward, Hogan and Nelson, Dev. Biol. 98: 70-79, 1983). We have been examining these phenomena by patch clamping
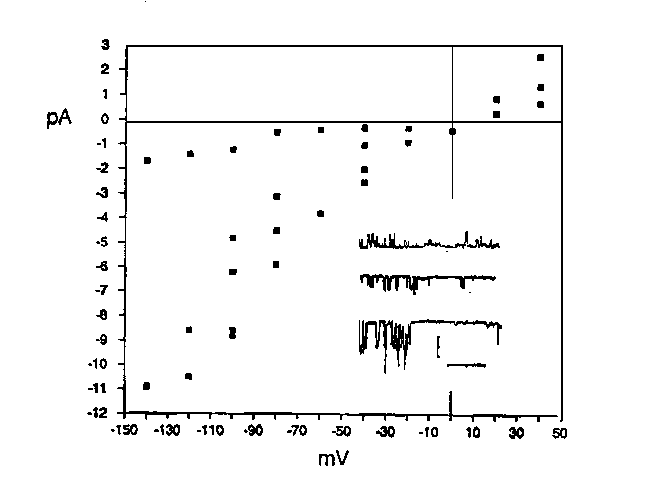
techniques. Spermatids were prepared from him-5 ( e1490 )males in Sperm Medium (SM; see Nelson and Ward, ibid.) and the patch pipette was filled with SM (for cell attached recordings) or various other solutions (for cell detached recordings). The small size (~5-6 microns for spermatids) of this cell required narrow tipped pipettes, which were fire polished so that the tips had an internal diameter of about 1 micron and had a resistance of 4-10 megaohm. Utilization of these narrow tipped pipettes allowed formation of high resistance (greater than 20 gigaohm) membrane seals to patch pipettes on both cell attached and broken cell detached (inside out) preparations, as measured on a List EPC-7 ,bandlimited at 1000 Hz by standard techniques (Wellis et al., Biophys. J. 57: 41-48, 1990). Cell attached patches, which have an area of about 5 microns (2) (by capacitive measurement), do not seem to affect cell viability for at least 30 minutes and can be obtained routinely on either spermatids or spermatocytes. Distinct levels of current appear at each applied voltage, indicating at least four types of ion channels in spermatid plasma membrane. In figure 1, the lowest conductance level observed is about 15 pS and the highest is 95 pS. These currents reverse between 10 and 15 mV negative to rest (arbitrarily set to 0 mV). A 200 pS ion channel is also occasionally observed (not shown in Fig. 1). These results indicate that it is feasible to study C. elegans sperm differentiation by patch clamping. Our first investigations will involve patch clamping spermatids and inducing their differentiation into spermatozoa while monitoring ion channel activities.