Worm Breeder's Gazette 12(5): 49 (February 1, 1993)
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
lin 25 is required for proper vulval cell fate determination (Ferguson and Horvitz. 1985. Genetics 110 17-72). In animals carrying null mutations in lin-25 , P6 .padopts a vulval fate whereas all the other VPCs adopt the 3° fate (Ferguson et al., 1987. Nature 326. 259-267 and our own observations). Iin 25 mutations recessively suppress the multivulva phenotype of Iet-60 (gf)and lin-1 (lf)mutations but not the multivulva phenotype of lin-12 (d)allele. In a previous gazette article we described our genetic characterization of lin-25 (WBG 11 No. 5, 77); here we report our progress towards cloning lin-25 and some preliminary results from genetic mosaic analysis.
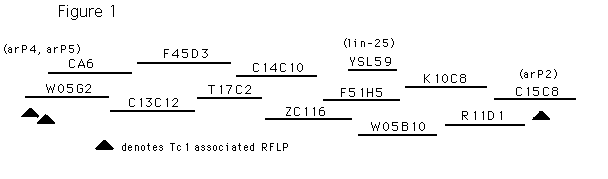
lin-25 cloning. We have identified a 10kb fragment that completely rescues the lin-25 mutant phenotypes (figure 1). When a plasmid containing 10kb subcloned from YL559 was injected at a concentration of 15mg/ml into animals homozygous for the temperature sensitive lin-25 mutation, n545 ,more than 80% of the F1 Rollers (n=100) (grown at the restrictive temperature) were rescued for the egg-laying defect. All the lines generated were also rescued. Deletion of 2kb from either end of the 10kb fragment abolished the rescuing ability.
lin-25 genetic mosaics. Temperature shift experiments indicate that lin-25 is required for the proper development of more than one component of the egg-laying system (Ferguson et al., 1987 and our own observations). We are scoring lin-25 mosaic animals, therefore, both for the vulval lineage defect and for the ability to lay eggs. To generate mosaics mutant for lin-25 in the AB lineage we constructed a strain of the genotype dpy-17 unc-36 ncl-1 ; unc-42 lin-25 (0); ctDp11 ( ctDp11 is-fusion between parts of chromosomes III and V; it complements an of these markers, Hunter and Wood. 1992. Nature 355. 551-555). We picked Unc NonDpy segregants, which were mosaics that had lost ctDp11 in AB or ABp. Half of them were Egl- (n=25). We have followed the vulval lineages in just two such mosaics so far (one AB mosaic and one ABp): both of these had wild-type vulval lineages and were Egl+. To generate P1 mosaics we constructed the strain sup-18 ncl-1 unc-36 ; unc-42 lin-25 (0); ctDp11 ; sup-10 ( n983 ).We picked wild-type segregants, which were mosaics that had lost c tDpl11 in P1 : sup-18 is a recessive suppressor of sup-10 ( n983 ),whose focus is in P1 .We have found two P1 mosaics so far; both of these were Egl-. There are several possible explanations for these results at the moment. Iin-25 might be required in two different tissues for proper egg-laying the VPCs (for proper fate determination) and a tissue derived from P1 (for some other, as yet unknown, aspect of egg-laying). The wild-type vulval lineages observed in the AB and ABp mosaics could be because of maternal rescue ( lin-25 homozygotes coming from a heterozygote are sometimes Egl+). A second possibility is that focus of lin-25 for proper vulval cell fate determination might be a tissue derived from both AB and P1 such as hyp7 .The focus for lin-15 for VPC fate determination is thought to be hyp7 (Herman and Hedgecock 1990. Nature 348. 169-171). We are currently examining the vulval lineages in more mosaic animals to distinguish these and other possibilities.
[See Figure]