Worm Breeder's Gazette 12(5): 32 (February 1, 1993)
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
The initial devclopment of the C. elegans embryo is characterized by
a stereotypical cleavage pattern: The first cleavage is unequal and
oriented on the anterior-posterior axis. During second cleavage, the
larger anterior cell, AB, divides equally and transversly to the
anterior-posterior axis. The Pl cell divides after AB and thc Pl
cleavage is unequal and oriented on the anterior-posterior axis (l;
Flg. lA). The orientation of the Pl division results from a rotation of
the centrosomes and nucleus that aligns them on the the same axis
as the previous division (2). We are interested in understanding the
mechanisms which specify this pattern. The previously identifiedpar
genes are required for the cytoplasmic localization of factors, some
of which act to control the cleavage pattern (3). We are ca~Tying out
a screen to identify new maternal effect lethal mutations that
disrupt the normal cleavage pattern.
L4 woqms were mutatgenized with EMS and their Fl progeny picked
singly to plates. Of 3073 Fl worms screened to date, 442 segregated
F2 maternal effect lethals. In 198 maternal effect lines, mutant
hermaphrodites p~oduced embryos that arrested with large numbers
of cclls, but little or no morphogenesis. Embryos from those lines
were filmed using time-lapse video microscopy to identify
mutations affecting the cleavage pattern. Three mutadons with
phenotypes resembling those of par mutations were recovered, as
wcll as dght mutations with novd phenotypes. Based on mapping and
complcmentation data, these dght mutations represent fivc or more
genes. At least three of the mutations appear to specifically affect
cleavage onentations.
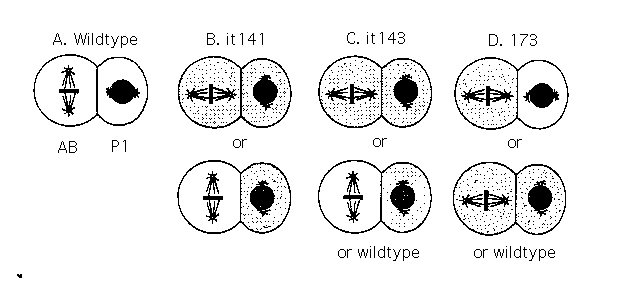
Hermaphrodites homozygous for itl41 (LGIV, between unc-22 and
dpy4 )produce embryos in which the earliest visible defect is the
orientation of second cleavage spindles. In all embryos, the Pl
division is transverse to the antenor-posterior axis. In about half
the em~ryos, the AB division is also incorrectly oriented, on the
anterior-posterior axis (Fig. lB). Ihe relative timing of ~e AB and Pl
cleavages is nomlal, however. In addition, P granules are localized
normally prior to the first and second cleavages. P granules are
prescnt in two cells at the four cell stage, presumably due to the
incorrect orientation of the Pl division. Third cleavage oricntations
are also abnormal.
The itl43 mutation causes a similar but more variable phenotype:
Embryos from itl43 homozygous mutant mothers appear normal up to
the second cleavage, at which time both the Pl and AB orientations
can be incoTrect ~Fig lC). Some embryos look normal at the second
cleavage, but then show altered orientations at third cleavage. This
allele complements itl41 and maps to LGI between dpy-S and unc-13 .
Hermaph~dites homozygous for mutation 173 produce many embryos
in which both AB and Pl divide on the anterior-posterior axis, but
with the normal timing. The remaining embryos show either the
reversal phentoype described above (AB dividing on the anterior-
posterior axis, Pl dividing transversly), or they look normal at
second cleavage but have third cleavage abnormalities (Fig. lD).
Onc complementation group consists of two alleles which were
identified because the Pl cleavage is transverse to the anterior-
posterioT axis. This phenotype is probably an indirect effect of
oogenesis defects, however. In mutant hesmaph~odites, oocytes are
produced from both sides of the proximal gonad (instead of from only
the outer side). This results in a double r~w of oocytes which are
about half the size of wildtype. After fer~lization, multiple female
pronucld are often visible and ccll cycles are slower.
We are furdur charactenzing the mutations that appear to
specifically affect cleavage crientation. We will determine if the
altered cleavage onentalions are due to failures of rotation of the
nucleus and centrosomes in Pl, accompanied by rotation in the AB.
Also of interest is the bchavior and fate of the daughtÇ cells derived
from an incorrecdy oriented Pl division. Fmer mapping of the
mutations, and analysis of the phenotypes of mutations in ~ans to
dcficiencies is in progress.
Figure 1. Schematic representations of wildtype and mutant
cleavage patterns. Centrosomes, microtubules, and DNA are indicated
by circles, lines, and shading. Cells that show incorrect cleavage
orientations are stippled.
2. Hyman and White, 1987. JCB 105:2123-2135.
3. Kemphues et al., 1988. Cell 52:311-320.