Worm Breeder's Gazette 12(4): 52 (October 1, 1992)
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
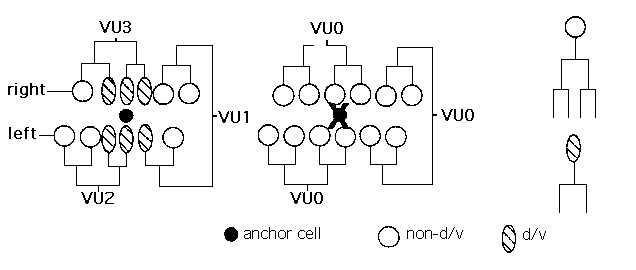
The cells of the ventral portion of the C. elegans hermaphrodite uterus result from divisions of three precursors, W1 , W2 ,and W3 (Kimble and Hirsh, Dev. Biol. 70: 396-417; 1979). Each W cell generates slightly different sets of intermediate precursors of differentiated progeny. One intermediate precursor produced by the W cells (d/v) divides dorsal-ventrally and undergoes one less round of division than the other precursor types. The three W lineages differ in the number of d/v's they produce (VU1, 2 and 3 produce one, two, and three d/v's respectively). Four bipotential cells generate the 3 W cells and the anchor cell; Z1 .pppand Z4 .aaacan each become either an anchor cell or VU3 ,and Z1 .ppaand Z4 .aapcan each become W1 or W2 .Kimble has demonstrated several types of regulation among these cells. Ablation of the presumptive anchor cell results in its replacement by the alternative precursor (which would otherwise become W3 ).Ablation of W3 results in W1 adopting a W2 lineage, while ablation of W1 leads W3 to adopt a novel lineage or change its polarity (Dev. Biol. 87: 286-300; 1981).
Results from our ablation experiments suggest that the anchor cell may be necessary for the d/v fate. We ablated the anchor cell during L2 lethargus/ early L3 ,after the time during which anchor cell regulation can occur. We found that all three W cells now follow a "VUO" lineage, in which no d/v's are produced, i.e. all cells undergo the fourth round of division characteristic of non-d/v's (13 VU lineages followed in 6 animals). In presumptive W1 and W2 lineages, all cells appear indistinguishable in terms of size and timing of division. However, in the presumptive W3 lineage, the cells that would normally be d/v's divide later than other cells. When the ablations were done after the first W division (during L3 ),approximately half of the presumptive d/v's underwent an extra round of division (3/3 animals). Therefore, cell fate is not irrevocably determined at the level of the W precursors.
We have determined the ventral uterine lineages in highly penetrant vulvaless mutants of let-23 ( sy97 )and lin-3 ( n3781 n1059 heterozygotes). In neither case was there a defect in d/v fate specification (4/4 animals for each strain). Thus the anchor cell does not appear to use the identical signal transduction pathway in inducing VPCs and d/v's. Furthermore, since d/v's are present in Vul mutants, the W0 lineages observed in anchor cell-ablated animals are presumably not an indirect consequence of the absence of vulval tissue. We are examining animals with VPCs ablated to confirm this result.
We infer that a signal from the anchor cell induces specialization of the ventral uterus. We are currently determining the ventral uterine lineages in lin-12 (d), lin-12 (0),and additional mutants to identify potential signaling molecules. We also plan to screen for mutants defective in specification of the d/v fate. [See Figure]
Dev. Biol. 87: 286-300; 1981.