Worm Breeder's Gazette 12(4): 44 (October 1, 1992)
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
mec-3 ,a homeodomain-type transcriptional regulator, is the primary determinant of cell type for three sets of mechanosensory neurons in C. elegans. mec-3 is of special interest because it is expressed at the end of a stereotyped cell lineage, and only in one of the two daughters or grand-daughters of unc-86 -expressingcells upon an asymmetric cell division. One property of the stereotyped cell lineages is that with every cell division, there is a change in cell fate. To account for this, we have postulated that in a stereotyped cell lineage, an ordered series of specific transcription factors cooperate with a cell cycle-specific transcriptional regulator to sequentially activate each other. Asymmetric cell division and cell cycle-dependent changes in gene expression are together sufficient to generate a stereotyped cell lineage. Understanding why mec-3 is well regulated should help us understanding these processes.
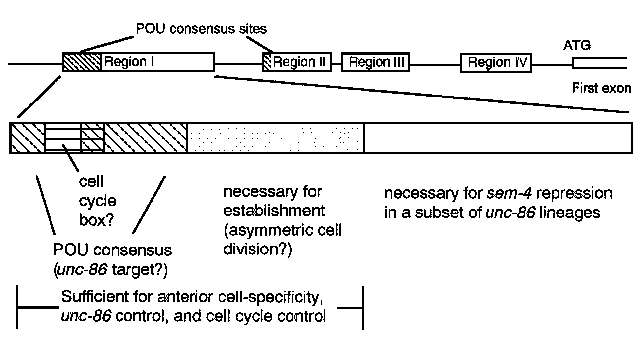
The promoter region of mec-3 has been studied by using mec-3 -lacZfusion and HS- mec-3 fusion promoter (Way et al., Genes & Dev. 5, 2199-2211). Various regions of the mec-3 promoter have been fused with a truncated heat shock promoter (leaving one heat shock repeat and TATA) followed by lacZ. The expression of lacZ is then tested after heat shock.
When a DNA fragment containing regions I, II, and III (defined by similarity with the C. vulgariensis mec-3 promoter) is fused with the truncated heat shock promoter, B-galactosidase is expressed only in the ten mec-3 -expressingcells. No detectable b-galactosidase is expressed without heat shock. When only region I is fused with the truncated heat shock promoter, b-galactosidase is expressed only in the postembryonic mec-3 -expressingcells (AVM, PVM, and PVD neurons) after heat shock and only when the cells arise, which means region I is sufficient to direct establishment but not maintenance synthesis of mec-3 .The mec-3 and heat shock promoter elements are insufficient by themselves to induce transcription but appear to cooperate when both are active. We are dissecting this 71 bp region I and have found that when a 29-bp subregion is fused with the truncated heat shock promoter, lacZ is expressed in all anterior daughters or grand-daughters of unc-86 -expressingcells, apparently including the PHC cells. PHC is an exception among the anterior descendants of unc-86 -expressingcells, in that it does not express mec-3 .However, PHC expresses b-galactosidase in a sem-4 ; mec-3 -lacZstrain (M. Basson and H.R. Horvitz, pers. comm.; J-Q. Run and J. Way, unpub.). These results suggest sem-4 is responsible for the inhibition of mec-3 in PHC. The second part of the region I might be the sem-4 binding site. sem-4 has been cloned by Michael Basson and is a zinc finger-containing protein.
Further mutation of the 29bp region leads to significant decrease or elimination of expression. Thus this 29-bp appears to contain targets for 1) unc-86 action, 2) cell cycle regulation as defined by the fact that only progeny of unc-86 -expressingcells express the fusion, and 3) asymmetric expression, as defined by the fact that only anterior daughters or grand-daughters express the fusion. This region includes, in the first half, a POU-domain consensus binding site and a core cell cycle box (CGMA) defined by the common sequences from yeast HO and histone H2 Bpromoters, and a novel sequence in the second half.
Our first-order speculation about the subdivision of sites in this region is that the POU consensus sequence is the unc-86 binding site, the CGMA would mediate cell cycle control, and the novel sequence could mediate asymmetric expression of mec-3 .Other arrangements are possible: for example, since the POU protein Oct 1st mediates cell cycle regulation of histone H2 Bin mammalian cells, the POU consensus site might have this function in mec-3 .However, because any protein binding to this region should be interesting, we plan to isolate binding proteins from C. elegans extracts using the standard band-shift assay. [See Figures 1&2;]
M. Basson and H.R. Horvitz, pers. comm.; J-Q. Run and J. Way, unpub.