Worm Breeder's Gazette 12(4): 36 (October 1, 1992)
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
mec-4 encodes a protein that is required for the mechanosensory function of the six touch receptor neurons. The deduced amino acid sequence of this protein includes two hydrophobic domains, each theoretically capable of spanning the cell membrane, and two Cys rich domains, the more C-terminal of which shares some sequence similarity with a Cys-rich domain in the LDL receptor. Because mec-4 appears to lack an N-terminal signal sequence, we predict that mec-4 may be situated in the membrane with N- and C-termini located inside the cell and most of the protein, including the Cys-rich domains, located outside the cell. We suspect that these Cys-rich domains, which are conserved among various degenerin genes, may be important for maintaining structure of mec-4 or for interacting with other proteins.
Rare dominant mutations in mec-4 induce swelling and lysis of the six touch receptors. Because mec-4 dominant alleles appear to alter osmotic balance in the touch receptor cells, we have suggested that mec-4 may normally function to regulate membrane permeability, perhaps as a subunit of a mechanosensory ion channel. The amino acid changes that transform mec-4 into a death-inducing protein are substitutions of large side-chain amino acids for Ala(442), which is situated adjacent to the second membrane spanning domain.
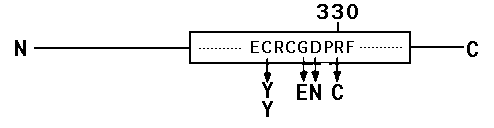
In order to identify the elements of mec-4 that are important for normal function, we have been sequencing the direct PCR products of genomic DNAs of EMS-induced recessive mec-4 alleles. In our sequence analysis of exon 8 of 31 mec-4 (r)alleles, we have identified a cluster of point mutations that fall within the second Cys-rich region of mec-4 .The amino acids that are affected are identical in Deg-1 and are also highly conserved in other degenerins. [See Figure]
Our sequence analysis of exon 8 of mec-4 ( u56 )helps explain a paradoxical observation. When we sequenced exon 12 we found that u56 encoded a substitution of Asp for Ala at position 442 in the theoretical protein deduced from the partial mec-4 cDNA clone. We had previously constructed an allele encoding this amino acid substitution through site-directed mutagenesis and observed that mec-4 (A sp442 )induced degeneration of the touch receptor neurons in transformed lines. However in the EMS-induced mutant, no degenerations were observed and the mutation was recessive in genetic crosses. The identification of a second mutation in cis in u56 makes it likely that the amino acid substitution at position 330 serves to block the ability of the Asp(442) to cause degeneration.