Worm Breeder's Gazette 12(4): 22 (October 1, 1992)
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
In C. elegans the pre-mRNAs of some genes receive the spliced leader SL1 while others receive SL2 . SL1 is trans-spliced onto conventional monocistronic pre-mRNAs containing an "outron" (Conrad et al.,1991, MCB 11:1921). SL2 ,however, appears to be specialized for trans-splicing onto the pre-mRNAs of downstream genes in polycistronic transcription units (i.e. operons) (see WBG 12:3, p. 72). We've been using the gpd-21 / gpd-3 "operon" to study SL2 -specifictrans-splicing. Ralph Hecht kindly provided us with genomic sequence surrounding gpd-2 &3and; pointed out the presence of an open reading frame upstream of gpd-2 (Lee et al., 1992, Gene: In press). We have now shown by RT-PCR and the isolation of cDNA clones that this ORF represents a real gene. It has been named mai-1 (for mitochondrial ATPase inhibitor) because of its homology to a mammalian gene which specifies a nuclear-encoded subunit of the mitochondrial ATP synthetase (Interestingly, the worm version doesn't appear to begin with a mitochondrial import peptide, as the mammalian gene does.)
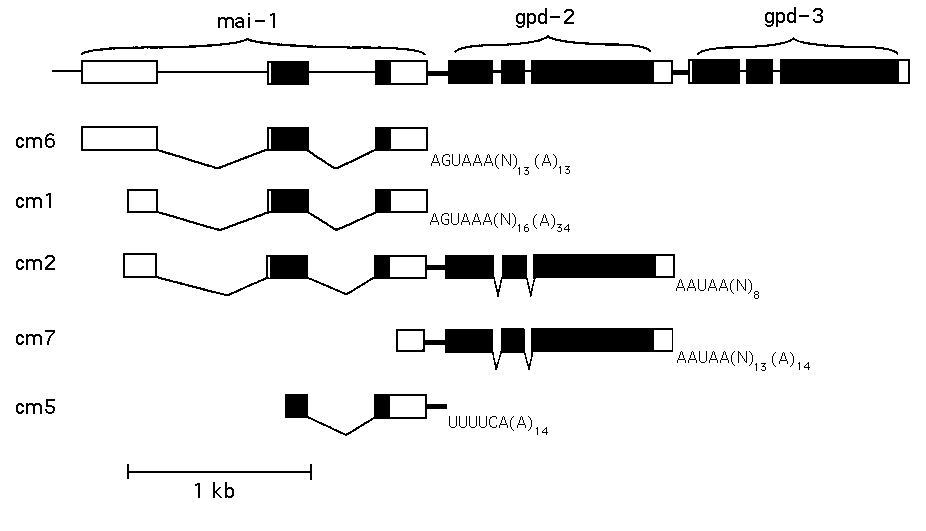
The structure of mai-1 ,shown in the top line of the figure, has been determined from genomic and cDNA sequences. Introns and exons are indicated in the traditional fashion with the coding portions of exons shaded. The intercistronic spaces between mai-1 and gpd-2 ,and between gpd-2 and gpd-3 are represented by narrow solid bars. We selected six cDNA clones using a fragment of genomic clone containing the second intron and third exon of mai-1 .The structures of the 5 types of clone are diagrammed below (clone cm1 was isolated twice.) Clone cm6 may be full length based on Northern analysis. The 3' ends of cm1 & 6 are just downstream of the unusual cleavage and polyadenylation signal, AGUMA. These two clones place the 3' end of mai-1 only about 100 bp upstream of gpd-2 ,suggesting that mai-1 could also be part of the gpd-21 gpd-3 operon. Additional evidence for this possibility is provided by clones cm2 & 7 which both contain part of mai-1 and all of gpd-2 on the same clone. In both clones, introns were properly spliced, and cm7 at least is polyadenylated at the normal site at the end of gpd-2 .However, the entire intercistronic space between mai-1 and gpd-2 remains intact in both clones, so the introns were removed but trans-splicing of gpd-2 didn't occur. [See Figure]
PCR experiments suggest that the abundance of clones from polycistronic RNAs in this cDNA library (provided by Bob Barstead) represents the relative abundance in the total RNA population. We attribute this abundance of an apparent RNA processing intermediate to the unusual mai-1 polyA signal (AGUAM) which may be an inefficient cleavage signal. We hypothesize that these cDNA clones representing the polycistronic pre-mRNA accumulate both because cleavage is slow and because trans-splicing of the resultant polycistronic pre-mRNA is a naturally slow event. Perhaps the most interesting clone is cm5 .Its 3' end is one base upstream of the trans-splice site for gpd-2 ,suggesting that the process of trans-splicing separated the polycistronic RNA, not the cleavage that normally precedes polyA addition. Furthermore, this clone is polyadenylated even though there is no MUAAA just upstream (the closest is the AGAUUU at the 3' end of mai-1 ,127 nt upstream).
Our results support the idea that mai-1 , gpd-2 and gpd-3 constitute a 3 gene "operon". Clones cm2 ,5 & 7 offer strong evidence for co-transcription of mai-1 and gpd-2 .Our model then predicts that gpd-2 should receive SL2 because it is a downstream gene in a polycistronic transcription unit. Although gpd-2 has previously been reported to be an SL1 -acceptor,we have performed two types of experiments (RTPCR with either SL1 or SL2 as upstream primers and primer extension following RNaseH cleavage with SL1 -or SL2 -specificoligos) indicating that some gpd-2 pre-mRNAs receive SL1 while others receive SL2 .This suggests the possibility that transcription of gpd-2 / gpd-3 might originate from two promoters. One would be upstream of mai-1 such that its transcript would include mai-1 , gpd-2 ,and gpd-3 .In this case gpd-2 would be expected to receive SL2 because it is a downstream gene. Clones cm2 ,5, and 7 indicate this configuration of mai-1 , gpd-2 transcription exists. A polycistronic RNA containing all three has not been detected, presumably because of the normal, efficient, cleavage signal at the end of gpd-2 .The other promoter may lie within mai-1 ,such that gpd-2 is at the 5' end of the transcript and thus it receives SL1 because it begins with an outron. Deletions of mai-1 in which only a part of the last intron and the last exon remain show good expression of gpd-2 and gpd-3 ,providing evidence for this transcript.
Clearly what we thought was a simple system to study SL2 -specifictrans-splicing (of gpd-3 )has become more complex because the "operon" actually is composed of three genes and multiple promoters.
WBG 12:3, p. 72.
Lee et al., 1992, Gene: in press.