Worm Breeder's Gazette 12(4): 12 (October 1, 1992)
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
Evolutionary relationships between species can be deduced from sequence divergence of proteins or DNA; however strains within a species generally show too little divergence to analyze in this way. The insertion and excision of transposable elements, however, are frequent enough events in mutator strains of C. elegans to allow the construction of cladistic trees. We have utilized a polymerase chain reaction (PCR) procedure designed for rapid chromosome mapping (1), to identify Tc1 elements which are conserved at the same insertion sites as initially determined in the Bergerac-BO strain, which contains 800-1000 copies of transposon Tc1 (2). Bands of varying size are generated in multiplex PCR sets, each containing an oligonucleotide primer from within Tc1 and several opposing primers chosen from chromosomal DNA adjacent to specific Tc1 insertions.
High- Tc1 strains RW7000 , DH424 , RC301 and TR403 were obtained from the Caenorhabditis Genetics Center (CGC, University of Missouri, Columbia); these constitute all of the CGC's strain holdings known to have high copy number for Tc1 .In order to minimize the risk of inter-strain contamination, worms were analyzed directly as received from CGC or after a single generation of propagation in our laboratory. Identical results were also obtained with stocks of RW7000 obtained previously from R. Waterston (Washington University, St. Louis, MO), of DH424 and TR403 obtained from P. Anderson (Univ. of Wisconsin, Madison, WI), and of DH424 obtained from T. Johnson (Univ. of Colorado, Boulder CO). We also tested a Bergerac-BO line obtained from CGC in 1988. The strains tested were geographically distinct isolates. Bergerac-BO was collected in Bergerac, France, before 1949; substrains of BO are also called EM1002 and RW7000 (3, 4). DH424 (also called EPC-4 )was isolated from soil in El Prieto Canyon, near Pasadena, CA in the 1970's (3-5). RC301 was isolated in Freiburg, Germany (5, 6); and TR403 was isolated in Madison WI (5). Previous reports (7) have classified these four high- Tc1 strains into 4 distinct groups according to the Southern-hybridization banding-patterns obtained with Tc1 probes. Strains BO/RW7000, RC301 and TR403 are mutators, while N2 and DH424 lack mutator activity at 20°C (4, 5, 8); of these strains, only RC301 is plg+ (7).
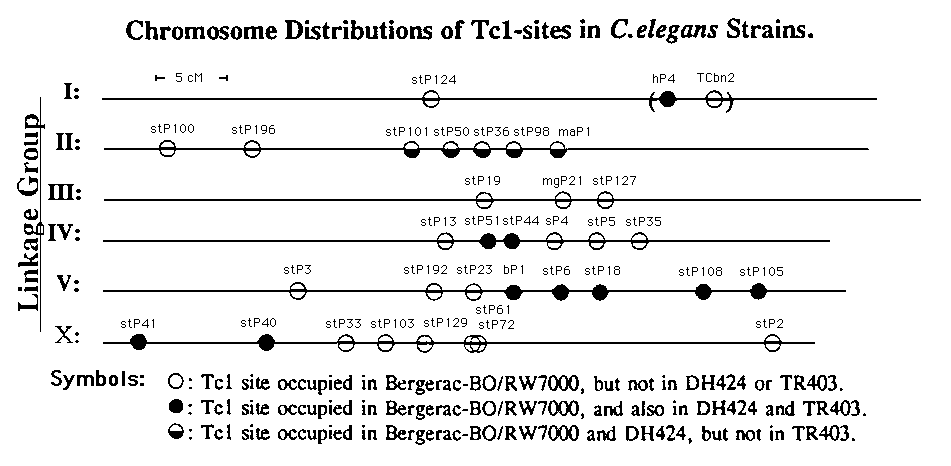
Adult worms, 1 per 15 µl of lysis mix, were processed as described by Williams et al (1), and an equal volume of 1.25 mM EDTA was added for storage. PCR reactions contained 2.5-5 µl of this solution (0.08-0.17 worm-equivalents) in 10 µl total volume. Bands indicating the presence of site-specific Tc1 elements, as characterized in strain RW7000 (1), were seen for all sites examined in Bergerac-BO and its subline, RW7000 .In strain DH424 ,however, 15 of 35 RW7000- Tc1 sites tested (43%) were occupied, while 10 of 35 (29%) were occupied in TR403 .Curiously, the sites occupied in DH424 comprised all 10 sites filled in TR403 ,plus an additional cluster of 5 sites in linkage group (LG) 11 which were vacant in TR403 .Another high-copynumber strain, RC301 ,lacked detectable Tc1 elements at all 35 loci tested, as did the low Tc1 -copy-numberstrain, Bristol-N2. When the chromosomal locations of conserved Tc1 sites were examined in DH424 and TR403 ,an intriguing pattern emerged: sites at which RW7000- Tc1 'sare conserved in these strains occur predominantly in clusters. For example, of 7 Tc1 sites mapped on LG-II in RW7000 ,all 5 sites retained in DH424 are contiguous, as are the 2 conserved sites (of 6 total) in LG-IV, all 5 conserved sites (of 8 total) in LG-V, and 2 (of 8 total) in LG-X. Except as noted for LG-II, these clusterings were identical in DH424 and TR403 .[See Figure]
If two high- Tc1 strains arose independently from low- Tc1 strains, each would be expected to have a different set of Tc1 insertion sites with little or no conservationÑas is indeed the case for RC301 vs. BO/RW7000, since no RW7000 Tc1 -specificbands were generated from RC301 DNA. Alternatively, two high- Tc1 strains might have arisen from a common progenitor strain, in which only a fraction of their present Tc1 sets had formed by transposition at the time of strain divergence. In this case, the "older" Tc1 loci would be shared by both strains, while those transpositions arising after strain separation would be unique to each strain and should be distributed at random with respect to the older sites. DH424 and TR403 retain 43% and 29%, respectively, of the RW7000 Tc1 markers tested; the value for DH424 is in good agreement with measurements indicating 300400 Tc1 elements in this strain (2). However, the observed distribution on the chromosomes, of conserved Tc1 loci, is far more clustered than expected by chance. The probability of finding at least as much clustering of conserved sites as found in DH424 is < 5 x 10-5, if they occur independently, while the corresponding probability for TR403 is 10-5. "CIustering" here refers to non-interspersion of the conserved and vacant Tc1 sites tested, as distinct from physical clustering of Tc1 sites along the chromosomes.
Such an outcome would not be at all improbable if the Tc1 loci in DH424 (and TR403 )did not arise independently, but instead resulted from a cross, either ancient or recent, between a Bergerac-BO-like strain and another strain with no shared Tc1 sites (e.g., RC301 or Bristol N2 ).Clustering would then reflect linkage between loci from the same parent, which was maintained once a hybrid lineage achieved homozygosity. The similarity of Tc1 -markerpatterns in DH424 and TR403 implies that both strains were derived from the same initial recombinant, which (if the cross preceded isolation of these strains) must then have colonized an extensive area of North America. Nevertheless, DH424 and TR403 are clearly distinct strainsÑDH424 has 5 Tc1 loci on LG-II absent from TR403 ,is non-mutator at 20°C (but active at 25°C) and ts-sterile (due to a zyg-12 mutation on LG-II), whereas TR403 is an active mutator at 20°C but is not ts-sterile (3-5, 8)Ñindicating genetic divergence or further outbreeding since their inception.
Analyses of fresh high- Tc1 isolates, from diverse locations, may help to resolve these issues; we would be pleased to receive such C. elegans cultures to test for conserved Tc1 sites.
[Supported by NIH grant RO1 AG09413 ]
2. Egilmez NE, unpublished observations, by quantitation of "dot blots" probed with Tc1 sequences.
3. Liao L, Rosenzweig B, & Hirsh D, Proc.Natl.Acad.Sci. USA 80:3585-89, 1983.
4. Eide D & Anderson P, Proc.Natl.Acad.Sci.USA 82:1756-60, 1985; Eide & Anderson, pers.comm.
5. Caenorhabditis Genetics Center (Columbia MO), strain notes.
6. Cassada R, WBG 9(3):29, 1986.
7. Katzenberg D, and Emmons S, WBG 7(2):34, 1980; Hodgkin J, WBG 10(2):140-141, 1988; Hodgkin J, WBG 11(5):60, 1991.
8. Moerman DG, and Waterston RH, Genetics 108:859-877, 1984.