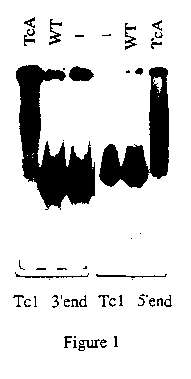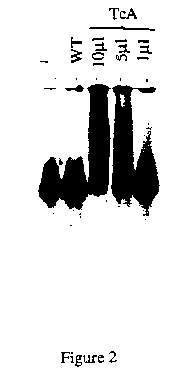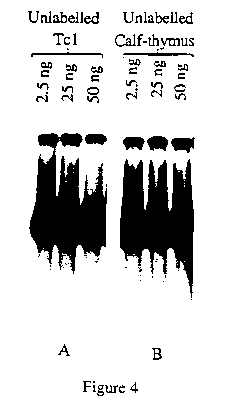Worm Breeder's Gazette 12(3): 90 (June 15, 1992)
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
The TcA protein is probably one of the proteins essential for Tc1 transposition. In order to study the Tc1 transposition mechanism biochemically, we would like to develop an in vitro reaction system. As an initial study of this problem, we have employed TcA protein overproduced in insect cells in DNA binding experiments with different parts of the Tc1 sequence.
Previous results from "South-Western blot" experiments indicated that TcA has a high non specific affinity for DNA both single stranded and double stranded (Schukkink and Plasterk, 1990, N.A.R. 18, 895-900).
To further characterize specific and non specific binding of the TcA protein, gel-retardation assays were performed with a variety of Tc1 fragments and calf-thymus DNAs.
These fragments, terminally labelled and incubated with nuclear protein extracts from insect cells infected either with recombinant baculovirus or with wild-type baculovirus (subsequently abbreviated as TcA- and WT- extract, respectively), were individually tested for formation of complexes with the TcA protein.
Figure 1 shows the results obtained with fragments covering 300 and 170 bp from the 5' and the 3' terminus ends of the element, respectively . A slowly migrating, diffuse complex is formed upon incubation of these fragments with the TcA-extract, but not with WT-extract . The intensity of the diffuse complex increases in a dose-dependent manner with TcA protein A (Fig. 2). Such a complex is also formed between the TcA-extract and non specific target (calf-thymus), indicating a significant non-specific affinity for DNA (Fig.3).This result was already observed for other eukaryotic transposases like the one from the P element of Drosophila melanogaster (Kaufman et al. 1989, Cell 59, 359-371).
To test for the specificity of the diffuse complex, unlabeled homologous and non-homologous competitor DNA fragments were included in the binding reactions with the labelled Tc1 5' terminus. Addition of an =10 to 200-fold excess of unlabeled Tc1 5' terminus fragment competes efficiently with the formation of the homologous complex (Fig.4). On the other hand the admixture of a similar excess of unlabeled calf-thymus does not interfere as well as the unlabeled Tc1 5' terminus fragment, indicating that TcA has a less good affinity for DNA other than Tc1 (Fig.4).
This results indicate that, despite its relatively strong non specific affinity for DNA, the putative transposase of Tc1 exhibits a better affinity for its Tc1 specific binding sites.
Kaufman et al. 1989, Cell 59, 359-371)