Worm Breeder's Gazette 12(3): 78 (June 15, 1992)
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
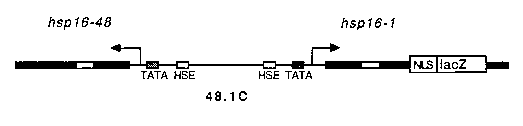
We have been studying the temporal and spatial expression of the hsp 16 genes of C. elegans by promoter analysis of transgenic animals carrying hsp16 -lacZfusions. Recently, stably integrated strains were derived from previously described strains carrying extrachromosomally inherited hsp 16 lacZ transgenes (Stringham et al., (1992) Molec. Biol. Cell, 3:221-233) by gamma irradiation mutagenesis (Jeff Way, personal communication). When animals carrying the integrated transgene, 48.1 C, are heat shocked and assayed for ß-galactosidase activity, blue staining is observed in most, if not all, somatic tissues. In contrast, animals which have not been subjected to a heat treatment do not stain at all. Recently, the inducible hsp16 promoters have been employed to achieve tightly regulated expression of various genes in C. elegans including mab-5 (Salser and Kenyon), and mec-3 (Way and colleagues).
We have been investigating the feasibility of inducing the heat shock response in individual cells of the worm by directing a laser microbeam to a particular target in an animal carrying an hsp16 -lacZtransgene, 48.1C, and then staining for ß-galactosidase activity. The intensity of a uv laser beam typically employed for ablation experiments was reduced by placing 14-20 mm of stacked microscope slides in front of the laser to act as a filter. This allows the manipulator to administer continual pulses from the laser to the target over a period of several minutes without killing the cell. Using this technique we have achieved single cell expression of ß-galactosidase in hypodermal, intestinal, neuronal, pharyngeal and muscle cells.
These results demonstrate that a uv laser microbeam normally employed for cell ablation can be adapted to produce a sub-lethal heat shock response in a given cell. The molecular basis for induction by the laser remains to be defined. The common feature of agents or conditions which induce the stress response is thought to be their ability to denature proteins. Laser irradiation might act to damage proteins directly by heating or indirectly by generating free radicals which could result in oxidative damage.
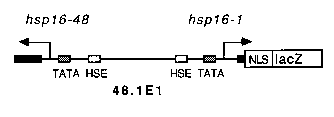
Transcription and translation of lacZ appeared to be unhindered by the laser treatment. Thus synthesis of functional proteins encoded by other genes under control of the hsp16 promoter should also be laser inducible in individual cells, offering unique opportunities to the C. elegans investigator who wishes to study cell-cell interactions. To take advantage of the bidirectional nature of the hsp16 promoter, we are currently inserting a polyclonal region into the 5' end of 48.1E1 such that expression of any coding region of interest can be conveniently coordinated with levels of lacZ expression. In this fashion, successful induction of the test transgene in a single cell can be verified by staining for lacZ expression. Alternatively, an integrated hsp16 -lacZtransgene could be crossed into a strain already carrying an hsp16 - fusion of interest. We are currently mapping the sites of array integration in our stable lines to facilitate in this endeavor.
We wish to thank Cathy Rankin for use of her laser and Steve Wicks for many helpful discussions. This research was supported by grants from MRC Canada, B.C. Health Research Foundation, and Stressgen Biotechnologies Corp.