Worm Breeder's Gazette 12(3): 72 (June 15, 1992)
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
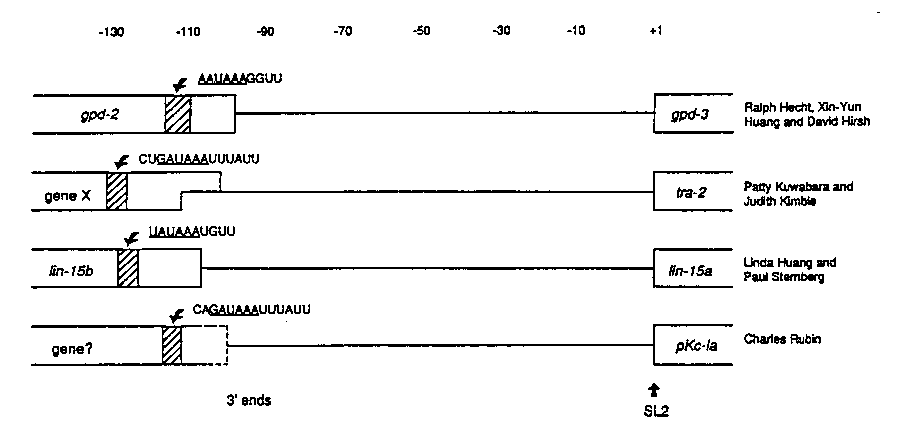
Why do the pre-mRNAs of some genes receive SL1 ,while others receive SL2 ? One hypothesis, proposed at the last worm meeting (See also WBG 12#1 p.43), states that any gene with an outron will receive SL1 ,but that SL2 trans-splicing is specific for downstream genes in polycistronic transcription units. The full length polycistronic RNA probably would never exist because polyadenylation and cleavage of the upstream gene would occur before transcription of the downstream gene would have proceeded very far. This cleavage is likely to leave the downstream RNA uncapped and this uncapped mRNA might be the signal for SL2 trans-splicing. This hypothesis makes several specific predictions. For instance, SL2 accepting genes should lie just downstream of another gene oriented in the same direction. Likewise, the promoter for an SL2 -acceptinggenes should lie upstream of the gene that precedes it. If polyadenylation is the event that separates members of a polycistronic unit, then interfering with cleavage and polyadenylation should lead to accumulation of a full length polycistronic pre-mRNA.
This hypothesis originated from the observation that the 5'-end of gpd-3 is only 99 bp downstream of the 3'-end of gpd-2 .However we now know of three other SL2 -acceptinggenes [See Figure 1] which appear to be about 100 bp downstream of another gene oriented in the same direction, strongly supporting the hypothesis and also suggesting that this 100 bp spacing may be mechanistically important. tra-2 is downstream of an, as yet, unidentified gene. lin-15 A receives SL2 and is just downstream of lin-15 B. The gene encoding Protein Kinase C(IA) receives SL2 ,and using primers constructed from sequence provided by C. Rubin, we have now shown by PCR and Northern blots that this gene is about 100 bp downstream of the 3' end of a previously unknown gene (a search of Genbank with the sequence of the 3' end of this genes did not show any significant homologies)
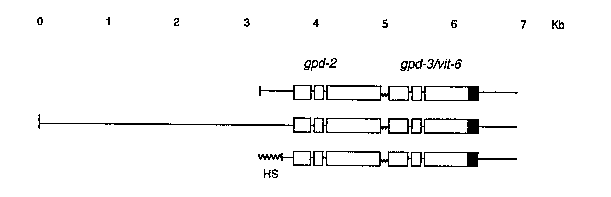
In order to test the prediction that the promoter for an SL2 -acceptinggene lies upstream of the gene that precedes it, we have made transgenic worms with genomic clones containing gpd-2 and a marked gpd-3 [See Figure 2] to determine how much DNA is needed for expression of gpd-3 . Even if we included all of gpd-2 and sequences 539 bp upstream of gpd-2 we were unable to detect any gpd-3 expression. However if we included an additional 3.5 kb of upstream sequence then we obtained a high level of gpd-3 expression, suggesting the gpd-3 promoter or enhancer lies well upstream of gpd-2 .We also inserted the promoter from the heat shock gene hsp-1641 142 bp upstream of gpd-2 in place of the gpd-2 upstream sequences. In this case mature gpd-3 mRNA trans-spliced to SL2 was made, and its synthesis was completely heat inducible. This demonstrates that when a polycistronic RNA is created artificially, it is capable of yielding mature, correctly-spliced mRNA [See Figure 2].
When we altered the AAUAAA polyadenylation signal downstream of gpd-2 (in the heat-shock construct) by site-directed mutagenesis, a band on Northern blots the size predicted for a polycistronic gpd-21 gpd-3 RNA accumulated following heat-shock. This polycistronic RNA was detected with probes to both the marked region of gpd-3 and to the intercistronic region which is not found as a part of mature gpd-2 or gpd-3 mRNAs. These experiments all support the idea that gpd-3 is transcribed from a promoter upstream of gpd-2 and that the cleavage/polyadenylation reaction of gpd-2 is involved in maturation of gpd-3 mRNA. Based on the strong correlation between SL2 -acceptanceand this unusual chromosomal arrangement, as well as our experimental results, we now feel reasonably confident that gpd-2 and gpd-3 ,and presumably the other SL2 -acceptinggenes, are transcribed as polycistronic units.