Worm Breeder's Gazette 12(3): 62 (June 15, 1992)
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
Ubiquitin plays key roles in a variety of cell functions. including selective protein degradation, DNA repair, cell cycle progression and chromatin structure. One major function of ubiquitin is to "tag" abnormal and short-lived proteins for recognition by a protease complex. This process is catalyzed through ubiquitin conjugating enzymes (E2's).
We have isolated and partially characterized two E2 genes in C. elegans. The complete cDNA and genomic clones were obtained by screening cDNA and genomic libraries using a PCR fragment amplified using primers derived from conserved regions of yeast and Drosophila E2 genes. One of these genes, named ubc-2 ,encodes a protein of 147 amino acid residues which is 95% identical to the Drosophila DUC-1 ,and 85% identical to S. cerevisiae UBC-4 and 5. The latter are believed to play major roles in degradation of abnormal and short-lived proteins, and are required for cell survival under stress conditions. The nematode ubc-2 can functionally substitute for UBC-4 and 5 in a yeast ubc-4 ubc-5 double mutant.
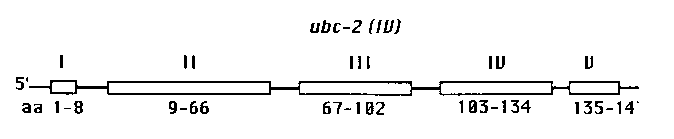
The ubc-2 gene has been physically mapped to chromosome IV in C. elegans. It consists of 5 exons divided by 4 introns and encodes an mRNA of 1.0 kb which is constitutively expressed at all stages of C. elegans development. Both cis and trans splicing process are involved in the maturation of the mRNA.
Current studies are aimed at defining the regulatory elements, tissue distribution and biological roles of ubc-2 .
Supported by the Medical Research Council of Canada and the B.C. Health Research Foundation.