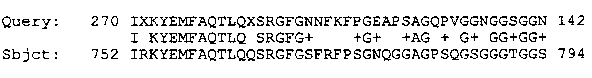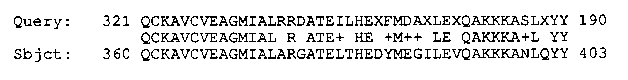Worm Breeder's Gazette 12(3): 60 (June 15, 1992)
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
The human tat binding protein ( tbp-1 )is a negative modulator of HIV transactivation which acts by binding to the Tat transactivating protein (Nelbock, et al, Science, 248: 1650-1653). Human tbp-1 shows similarity to a number of proteins involved in disparate functions in yeast and other organisms, which include the valosin containing protein and the yeast protein CDC48 p,which is involved in cell cycle control (Frohlich, et al, J. Cell. Biol. 114: 443-453 (1991)). While some regions of these proteins show a high degree of similarity to tbp-1 ,the sequence of tbp-1 is quite different from the other members of the family in the carboxyl terminal region.
We have isolated two cDNAs that are members of this gene family from an embryonic library. One of these, CEESH69 ,appears most closely related to the CDC48 ptype members of the family based on the sequence of the clone at the 3' end. However, the other clone, CEESA17 ,appears to encode a protein that is strikingly similar to tbp-1 ,including the carboxyl terminal region unique to tbp-1 .The alignments of both proteins are shown below. We are currently mapping and sequencing these clones and will be characterizing them in more detail.
Alignment of a translation of the EST from the 3' end of CEESH69 with the 806 amino acid porcine valosin containing protein (Koller and Brownstein, Nature 325: 542-545 (1987)) [See Figure 1].
Alignment of a translation of the EST from the 3' end of clone CEESA17 with the 404 amino acid tbp-1 [See Figure 2}.
Frohlich, et al, J. Cell. Biol. 114: 443-453 (1991)
Koller and Brownstein, Nature 325: 542-545 (1987)