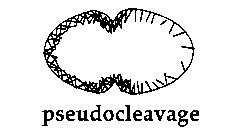Worm Breeder's Gazette 12(3): 112 (June 15, 1992)
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
The actin rich cortex of many animal cells forms a network subjacent to the plasma membrane that is capable of large scale movements in this plane. For example, in migrating fibroblasts and in nerve growth cones, the actin network continually flows away from the leading edge in a concerted manner at approximately 6um/min (1). It appears that monomeric actin is polymerised at the leading edge into filaments which join the network and then move away from this region. The filaments disassemble into monomers after moving away. A cytoplasmic flow in the opposite direction to the cortical flow (i.e towards the leading edge) could carry these actin monomers back to the leading edge to complete the cycle. The driving force for the movement of the cortical network could be gradients in myosin generated tension (2). If the mechanical tension of the network at the leading edge is lower than that elsewhere, then the network would tend to collapse back on itself towards the regions of higher tension. These gradients of tension could also power the compensatory cytoplasmic flow.
We have been looking at pseudocleavage in C. elegans as a possible and novel example of this cortical flow phenomenon. During pseudocleavage (PC), the anterior of the egg cortex undergoes a series of contractions whilst the posterior cortex remains relatively quiescent. Cytoplasmic material ( and possibly cytoplasmic determinants) flow from the anterior to the posterior, P granules become localized in the posterior (3), the pronuclei move towards each other (4) and a PC furrow forms and regresses. In addition, foci of actin become concentrated in the anterior of the embryo (5). Using the 4D time lapse system, we have analyzed the behavior of granules associated with the cortex during PC. In other systems, the movement of such granules parallels that of the underlying actin cytoskeleton (1). Pseudocleavage begins with waves of contraction moving from the posterior end to the anterior end at the same time as the anterior cortex begins to contract. These waves cease, and cortical granules stream from posterior to the site of the forming PC furrow. At the same time, cytoplasmic material flows in the opposite direction from anterior to posterior. The cortical flow occurs at a rate of between 3-7um/min which is consistent with previously determined rates for cortical flow. It is restricted to the granules in the posterior of the embryo: those in the anterior exhibit random movement. Thus, PC seems to involve a directed and continual movement of cortical material away from the posterior pole and a cytoplasmic flow in the opposite direction.
If a pronuclear stage embryo is treated with the microtubule polymerization inhibitor nocodazole, then no pronuclear migration occurs and a small spindle is formed around the centrosomes associated with the sperm pronucleus. This attenuated mitotic apparatus lies adjacent to the cortex at a variable position in the posterior part of the embryo. When such a drug treated embryo attempts to cleave, cortical material flows away from the region of the cortex next to the spindle at between 3-7um/min. At the same time, cytoplasm flows towards this region of the cortex and the anterior cortex contracts vigorously and repeatedly. A regressive cleavage furrow is formed between the zones of anterior contraction and posterior cortical flow just as in PC. These cortical and cytoplasmic flow patterns can also be seen in nocodazole treated AB and P1 blastomeres, again with the directions of the flows determined by the position of the attenuated spindle. In each case the flows and the position of the transient cleavage furrow mimic those seen during PC. This suggests that during PC some component of the sperm asters is inducing cortical material to move away form the asters, possibly by locally relaxing the mechanical tension of the actin network in the posterior part of the embryo. This may also occur during cytokinesis, with the asters of the mitotic spindle relaxing the cortical tension at the poles of the cell and hence inducing the network to flow into the equatorial region to form a cleavage furrow. A model for pseudocleavage is shown in Figure 1.
(2) White, J.G. & Borisy, G.G., J Theor. Biol. 101, 289 (1983)
(3) Strome, S. & Wood, W.B., PNAS (USA) 79,1558-1562 (1982)
(4) Albertson, D.G., Dev. Biol. 101, 61-72 (1984)
(S) Strome, S., J. Cell Biol. 103, 2241-2253 (1986)