Worm Breeder's Gazette 12(2): 92 (January 1, 1992)
These abstracts should not be cited in bibliographies. Material contained herein should be treated as personal communication and should be cited as such only with the consent of the author.
We are interested in studying the role of extracellular matrix components in cell migration. Type IV collagen, a prominent component of basal laminae, has been implicated in migration in vitro and in the neural crest (e.g. Halfter et. al., Dev. Biol. 32:14-25,1989). The opposite result, however, has been seen in the grasshopper limb, where initial axonal outgrowth is not affected by the enzymatic removal of the basal lamina (Condic and Bentley, Neuron 3:427-39,1989). We are interested in seeing whether type IV collagen could be involved in migration in C. elegans. Two collagen type IV genes, clb-1 ( let-2 )and clb-2 ( emb-9 ),have been isolated and cloned by Kramer and his colleagues (Guo and Kramer, J. Biol. Chem. 264:17574-82,1989). We decided to look at the migration of the Q cells and their descendants in mutants carrying temperature-sensitive alleles of these genes.
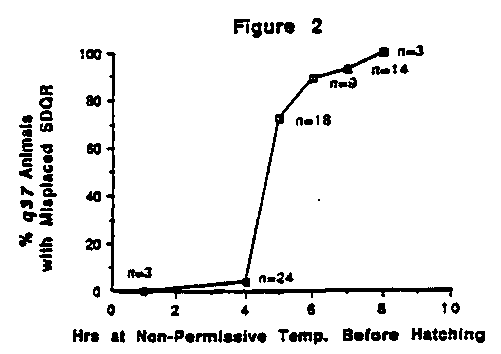
The Q neuroblasts are born late in embryogenesis between V4 and V5 .During L1 ,the Q cell on the left (QL) and some of its descendants migrate towards the tail while the a cell on the right (QR) and its descendants migrate towards the head (Sulston and Horvitz, Dev. Biol. 56:110-56, 1977). As a preliminary way of studying these migrations, we scored the final positions of the Q cells' descendants in let-2 ( q37 )and emb-9 ( mj70 )mutants shifted to the non-permissive temperature several hours before hatching. In both mutants, QR's descendants SDQR and, to a lesser extent, AVM were consistently located too far anterior (see figure 1) whereas QL's descendants seemed to be in the wildtype position. For let-2 ( q37 ),this migration defect was seen if the embryos were shifted to the non-permissive temperature at least five hours before hatching (see figure 2). The same phenotype was seen in emb-9 ( mj70 ) mutants shifted to the non-permissive temperature even one hour before hatching (n=29).
[See Figure 1]
The shift in position of SDQR and AVM is not large, but we find it interesting because it is so reproducible and because mutations in BOTH collagen genes lead to the same phenotype.
It is not clear from these results the mechanism by which collagen exerts its effect on cell migration. Does collagen (or another molecule that binds to collagen) act as a "stop sign" for these cells? Does collagen create a gradient along which the cells can migrate? Alternatively, are these effects on cell migration due to a secondary effect of collagen disruption? For example, is the extracellular matrix not as tightly woven, allowing cells to migrate faster through it? Whatever the mechanism, these results suggest that the basement membrane is involved in migration of the Q cell descendants.